 This is the first of a series of articles concerning the stone former phenotypes.
This is the first of a series of articles concerning the stone former phenotypes.
‘Phenotypes’ means literally the types of appearances of stone formers as observed medically, by which is meant observed using the common tests and measurements of medicine. These ‘types’ have value as they constellate abnormalities that might be individually confusing into patterns that can be recognized and that have implications for treatment and prognosis.
More precisely, medicine works entirely from abnormalities, meaning departures from what I might call the normative trajectory, the sum of all measurements and characteristics one expects among healthy people as they move through life. The abnormalities of medicine are all departures that produce a state of unwellness, or illness as is more accurately said. Having stones is an example.
Having stones and, as well, other kinds of abnormalities defines the individual phenotypes of stone disease. Here is the first one, the one seemingly most simple. It is, like all things we find in nature, not at all simple, but rather mysterious in its origins and uncertain in its exact boundaries. But medicine is a practical profession, not itself a science, and makes do with what it has, even when what it has is uncertain in its boundaries and an object of vigorous scientific debate.
Why Care To Define This Type of Patient?
The idiopathic calcium oxalate (CaOx) stone former (ICSF) is the most common kind of kidney stone patient at this time and in the Western Hemisphere. Most of you who are reading this site are one, or are a physician caring for one or more, or a scientist doing research on this kind of stone disease. The majority of treatment trials for stone prevention have enrolled mainly ICSF and therefore the results of such trials apply particularly well to them.
I mean to explore all of the main stone forming phenotypes, as it is time to do so, and what better place to start than with the one most common?
How is this Type of Patient Defined?
These patients are those whose stones are entirely calcium crystals, predominantly calcium oxalate, and not caused by any systemic disease. ‘Entirely calcium crystals’ means uric acid, struvite, cystine, and drug or rare organic crystals are not present in any stones.
There are three major kinds of calcium crystals in stones: calcium oxalate, hydroxyapatite, and brushite – calcium monohydrogen phosphate. This latter crystal is associated with a specific kind of tissue injury pattern in kidneys, so patients with brushite in any stone are excluded. Calcium oxalate can be present as the monohydrate or dihydrate (COM and COD) and are often reported in this way. I do not consider this distinction in the definition of ICSF. This distinction arises mainly from our own work, and historically brushite stones did not exclude patients from the ICSF category. The Mayo Clinic study to which our work is compared in this article also excluded brushite stone formers.
This leaves patients whose stones are composed of calcium oxalate and hydroxyapatite, with the former predominant.
How do we know a systemic disease is not a cause?
There are lists of the common ones, and screening is used to exclude them.
What about idiopathic hypercalciuria?
This trait is rampant among ICSF. We do not call IH a disease, but rather an otherwise benign genetic trait that by raising urine calcium raises risk of stones.
What Does ‘Predominantly CaOx’ Stones Mean?
In practice it means above 50% CaOx in all analysed stones taken on average. Since ICSF cannot by definition have uric acid, struvite, cystine, or drug or rare organic crystals in their stones, the other crystals they can have are forms of calcium phosphate (CaP): hydroxyapatite (HA) or brushite (BR). As I have just pointed out, because brushite stone formers have special peculiar features, ICSF are further defined as having no brushite in stones so the CaP is all hydroxyapatite.
Some time ago we took a close look at the fraction of CaP in stones from our calcium stone forming patients. 
These data are from all stones of 1201 patients who harbored no systemic disease as a cause of stones and whose stones contained CaOx and no uric acid, cystine, struvite or uncommon elements like drugs. For each patient we calculated the fraction of crystals that were calcium phosphate (CaP); the rest, given our exclusions, had to be CaOx.
But these were not ICSF, but idiopathic calcium stone formers because we included those with brushite in stones in order to compare HA and BR distributions.
For clarity we fitted the distribution of % CaP in stones from each patient as a continuous curve; the underlying data, remember, is the CaP% for a single patient.
At the left, the distribution peaks just above 2 % CaP meaning 98% or more CaOx.
The inset in the left panel uses a log transformation to expand the low range. A sizable peak of patients have less than 2% CaP (98% CaOx), and a huge fraction of patients have less than 10%.
This is clearly seen in the right hand panel. A large peak of patients have less than 20% CaP (Dashed line), with a peak at 10%; but the numbers of patients involved (compare the vertical axes of the right and left hand graphs) is a tenth of the total. The data for BR, as a component of CaP in stones show very low abundances until it becomes a predominant phase.
One can easily argue that more than 10% CaP (<90% CaOx) is uncommon, and that a CaOx fraction of 90% or more would in fact define the great mass of patients and enclose most of the peak in the left hand graph. Certainly, being most liberal, 20% CaP is an outer limit of the large first peak.
Because of our interests in the physiology of calcium phosphate stone formation, we used these figures to define a set of eight ranges for CaP%, and looking back I agree with our younger selves. But if I were to use these data to define a clear phenotype, 50% CaP or less would not be to my present taste. The purest group, in the first peak, would have an average stone CaP of <2%; a generous upper limit (the bottom of the huge initial peak) would be 10% – 20% CaP. So, perhaps, the field of stone practice and research might benefit from one of these two limits in place of the less than 50% CaP presently employed.
Which one would depend on what one is trying to do. For pure research, the first peak at 1%, ending around 2 – 3% would be ideal. For clarity in practice, perhaps 10% – 20% is best. Of these I would pick 20% as a most generous outer limit.
So, in practice, one excludes all stone components except CaOx and HA, and ideally chooses those with 20% or less HA as ICSF, if one follows my reasoning. Up until now, and perhaps in the future unless people agree with me, the 50% HA cutpoint was used, so it is the criterion for all of the work I review here.
How Do Their Stones Form?
We have described 30 ICSF in ways that permit us to offer an answer to this kind of question. We found that stones form as overgrowths on ‘plaque’, deposits of calcium phosphate in the kidney tissues.
They Grow on Papillae
The renal papillae are the terminal portions of the kidneys from which the final urine leaves into the collecting system. In ICSF stones are frequently observed growing on the papillary surfaces. When they are unattached, one can find remnants of papillary tissue on them as a sign of their origins.
They form on Plaque
This is a complex idea. We have a wonderful article on the site about plaque and how stones form on it, and this is a good time to look if you have not looked before and do not really know how stones form.
The article and its movies will tell you that plaque is interstitial – in between the tubules and vessels of the kidney tissue. This plaque is made of the same kind of 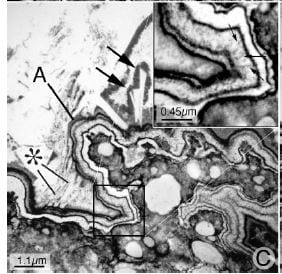 calcium phosphate as one finds in bone – hydroxyapatite. When the covering layers of the tissue give way over a deposit and it is exposed to the urine, crystals form over it because of supersaturations in urine, and layer by layer a CaOx stone results.
calcium phosphate as one finds in bone – hydroxyapatite. When the covering layers of the tissue give way over a deposit and it is exposed to the urine, crystals form over it because of supersaturations in urine, and layer by layer a CaOx stone results.
How they form their layers is illustrated in the enigmatic modernist picture that I used as the large featured image and took from our original paper. Here it is again, so you can see it as you read. The plaque is in the lower right corner; black is organic material, white is crystal. Plaque is in reality islands of HA crystals floating in an organic sea.
This plaque was exposed to the urine because the thin covering over it, the shiny outer layer of the papilla facing the urine, somehow gave way. The urine proteins coated the naked plaque; you can see this as a black layer over the plaque. In the upper right boxed inset a region of coverage is enlarged, and the thin black innermost organic layer clearly visible.
Because urine is supersaturated with calcium phosphate in most stone formers, crystals of CaP formed in the black covering layer (arrows point to the white appearing crystals), they were covered over, more formed, and eventually one has a ribbon of crystals and organic molecules. The letter A points to the ribbon. In the inset you can count the layers.
For reasons I do not know, the process stops its orderly ribbon making, and crystals just begin extending off of the surface to make the base of an enlarging stone (the * points to such extensions). All of the white sea upward and to the left is crystal, which is the stone mass near to the attachment site. Almost all of the crystal is still CaP. Further out, it becomes CaOx.
Is Formation on Plaque the Rule?
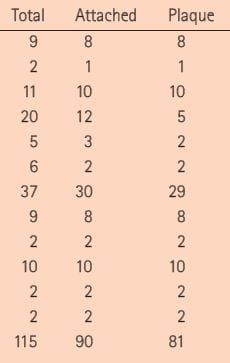 Plaque usually occupies no more than 5 – 15% of the papillary surface, but in a formal trial that counted whether stones being removed at surgery were on plaque or not they were indeed on plaque as opposed to the far more abundant plaque free surfaces. In this snippet from Table 1 of the published trial, each row but the last gives the number of total stones and the number attached, and attached to plaque; of the total of 115 stones 90 were attached and 81 were surely taken off plaque during surgery. The statistical odds of this from chance were negligible. The point estimate for growth on plaque was 73% of stones are forming on the 5 – 15% of papillary tissue that had plaque in it. The remaining stones, not attached, mostly had plaque attached to them, showing they had grown there.
Plaque usually occupies no more than 5 – 15% of the papillary surface, but in a formal trial that counted whether stones being removed at surgery were on plaque or not they were indeed on plaque as opposed to the far more abundant plaque free surfaces. In this snippet from Table 1 of the published trial, each row but the last gives the number of total stones and the number attached, and attached to plaque; of the total of 115 stones 90 were attached and 81 were surely taken off plaque during surgery. The statistical odds of this from chance were negligible. The point estimate for growth on plaque was 73% of stones are forming on the 5 – 15% of papillary tissue that had plaque in it. The remaining stones, not attached, mostly had plaque attached to them, showing they had grown there.
Plaque Builds up because of Altered Kidney Physiology
Plaque is not unique to stone formers. Kidneys from people who never formed a stone can have plaque in them, But ICSF tend to have a lot more plaque than non stone formers, so the amount on which stones can form is larger.
Why ICSF form more plaque than normal people is not known but theories are being pursued in experiments to this day. Our idea is that plaque arises because of idiopathic hypercalciuria, a very common trait in ICSF. We have suggested that increase of calcium delivery out of the earliest segments of the kidney tubules – the proximal tubules – sends excess calcium to later segments from which it enters the vessels that provide blood to the papillae. The extra calcium supersaturates the deep papillary tissues and leads to plaque.
Here is a moment of choice.
You can read on and I will tell you more of the details.
If you move on, take my word for it that we have details to tell about.
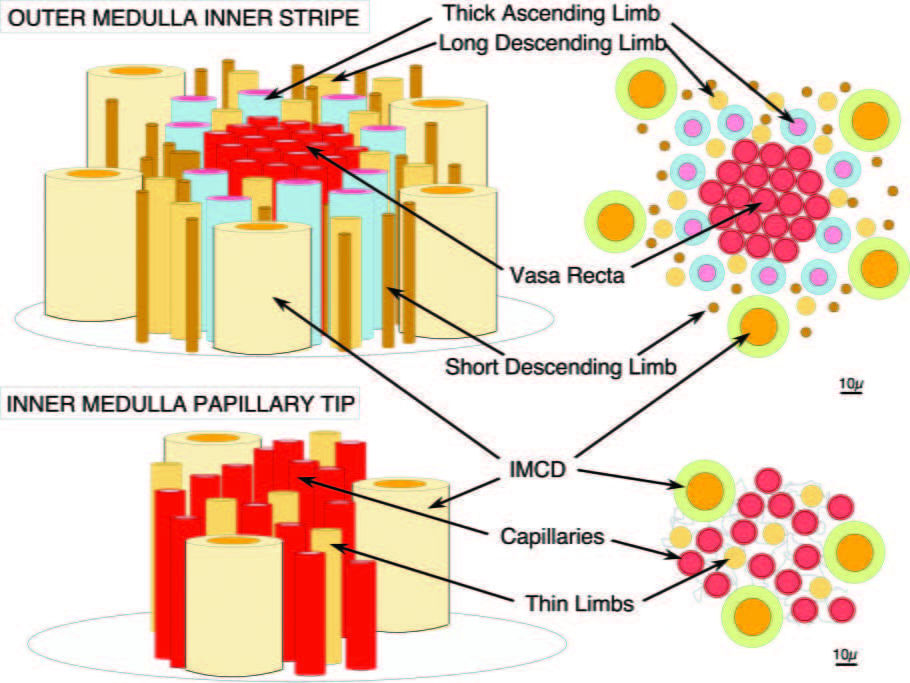
If you stay, take a look at the upper part of this picture which is from a recent review we published.
The kidney is shaped a little bit like a mushroom. The medulla is like the upper part of the stalk and the papillum like the end of the stalk where it fits into the ground. The cap is the cortex which we are not concerned with right now.
The inner stripe of the outer medulla would be up toward the cap and it is made of blood vessels (vasa recta, or straight vessels) and tubules: the descending limbs which are conveying tubule fluid downward into the papillum, and the thin and thick ascending limbs which are conveying fluid back up.
Here is the main point: The thick ascending limbs are arranged around the vessels in a ring – look at the cross section at the upper right. These limbs reabsorb calcium without water. That calcium can move into the descending vessels and be borne down into the papillary tip where plaque forms.
Now look at the bottom two panels. The plaque forms in the outer membranes (basement membranes) of the thin limbs, and these are surrounded by capillaries. The blood in these capillaries comes down from the bundles that have been enriched with calcium by the thick ascending limbs. So in everyone, patient or normal there is a tendency for the kidney to wash extra calcium down into the spaces around the thin limbs, increase saturation there, and foment plaque which is simply calcium phosphate crystals.
But idiopathic hypercalciuria has a special trait. People with IH tend to deliver more calcium out of the cortex into the descending thin limbs, which loop around and come back up as the thin ascending limbs that merge into the thick ascending limbs – these are essentially names for different regions of what are continuous hairpin shaped tubes.
If you deliver more calcium to the thick ascending limbs they can reabsorb it into the spaces around the vessel bundles, and enrich the down coursing blood more than is normal. This will promote supersaturation around the thin limbs and therefore promote plaque. We have proposed this idea and call it ‘vas washdown’. It is simply theory, but so far has been predictive.
What About Idiopathic Hypercalciuria?
We now understand from the work of Gary Curhan that urine calcium excretion is a graded risk factor and that stone risk rises above baseline at about 200 mg/day in men and women. If by hypercalciuria one means a urine calcium excretion high enough to cause a disease like stones, that threshold is 200 mg/day.
But Curhan and I would not advocate cutpoints, even so rational one as this appears to be. What he tells us is that risk rises pari passu from about 100 mg/day smoothly upward. At 200 mg/day the lower 95th percentile of the risk ratio compared to 100 mg/day passes above 1, meaning risk is almost certainly increased. So, there is no simple ‘hypercalciuria, merely upward ranging of risk as urine calcium rises above 100 mg/day, a value which many normal people exhibit.
My articles on idiopathic hypercalciuria and hypercalciuria present ample materials showing high urine calcium in stone formers, and there is no reason to add more here. The article on the fractions of phosphate in stones also has extensive data on urine calcium in stone formers. Put simply, they are as a group prone to ‘hypercalciuria’, to well above 200 mg/day or more of urine calcium excretion, and in ICSF it is always idiopathic – or else the patient would be otherwise named for the systemic disease raising urine calcium excretion.
Other Stone Risk Factors in ICSF
High urine oxalate, low urine volume, low urine citrate all occur in ICSF, and they have been the subject of prior articles on this site. LIke hypercalciuria they are graded risk factors, and cutpoints are no longer an ideal way to describe abnormalities.
The Turning of the Road
Our ICSF
If you read our papers, among the 30 ICSF we have published kidneys contain two kinds of calcifications: Plaque and stones. The stones are either attached to plaque, or found free in the urinary system. Of those found free, a majority have telltale plaque residues on them denoting a prior life as an overgrowth on plaque.
 But science is wonderful its twistings and turnings, and so the story is not at all finished but rather in a state of what I might call narrative suspension: No one really knows all of it, and therefore causes are mysterious and attractive as an old well.
But science is wonderful its twistings and turnings, and so the story is not at all finished but rather in a state of what I might call narrative suspension: No one really knows all of it, and therefore causes are mysterious and attractive as an old well.
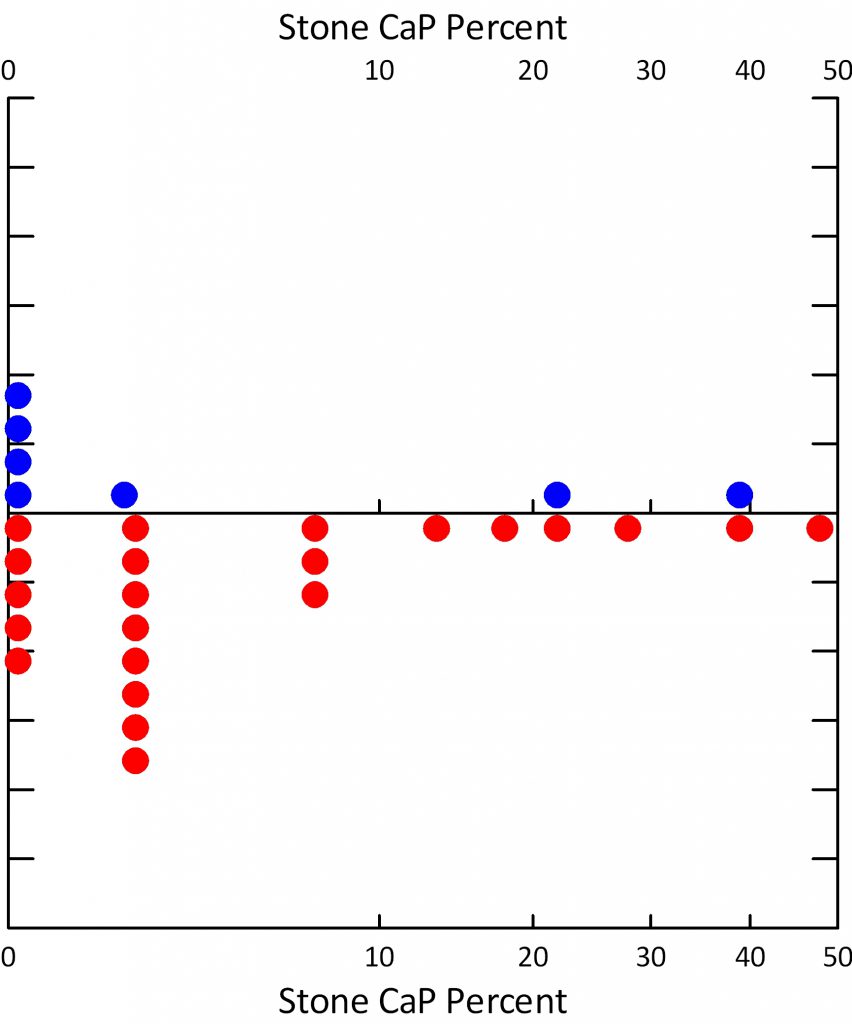
Our 30 ICSF were 22 males and 8 females. The average age was 52.
The fraction of stones with CaP ranged from 0 to just under 50%, with a sizable fraction below 10%. The women (blue) had on the whole even less CaP in stones than the men. Even by a stringent criterion of less than 20% CaP in stones most of these patients would have qualified as ICSF.
It is among these 30 patients we found only the two forms of calcifications, and so published in our papers.
Quantitatively, plaque averaged 8% of papillary surface, and an average of 10 stones was found on plaque per patient; urine calcium averaged 237 mg/day.
The Large Mayo Clinic Series
Dr Amy Krambeck and her colleagues published a large surgical series of patients at Mayo Clinic. Unlike our cases, her’s were unselected consecutive patients who came to her for stone removal via percutaneous nephrolithotomy – the type of procedure mainly used in our series by Dr James Lingeman – our surgical research colleague.
 In her series were 37 patients classified as ICSF, 70% female, and older (62 years) than ours. Among them she found rather modest amounts of plaque and some tubule plugging with crystals.
In her series were 37 patients classified as ICSF, 70% female, and older (62 years) than ours. Among them she found rather modest amounts of plaque and some tubule plugging with crystals.
The amount of plugging was modest among her ICSF with an average surface coverage of 0.3%, a frequency of about 28% of cases (graph at right, CaOx cases at right of the graph) and at least 1% in only 4 cases, but we reported no plugging and far more plaque.
Plugging is very common in stone formers, and we have reported it in almost all other phenotypes. As we found, she found extensive plugging in brushite, and apatite stone formers (CaP stone formers) and in patients with intestinal malabsorption and stones. But at the time of this publication we had not reported it at all in ICSF.
Plugs could be a second pathway for stone production because the open end of the plug often shows an overgrowth which could detach and grow or simply grow in situ into a clinically important stone.
So the differences between our series with respect to plugging are surprising and important.
The fraction of papillary surface covered with plaque was 3.6%, lower than we have found. Likewise urine calcium averaged only 210 mg/day, far lower than we have reported.
Where stones grew was not reported by Dr Krambeck, so we cannot compare her patients to ours in this respect.
One factor that probably accounts for the differences between ourselves and her group is that her cases were consecutive, ours selected as what appeared to be excellent examples of ICSF. Differences in surgical approach or technique are most unlikely in that Dr Krambeck worked in our group prior to moving to Mayo Clinic, and actually trained with Dr Lingeman in the specialized techniques used in this research.
The Small Mayo Clinic Series
A subsequent publication presented details of the ICSF group in isolation along with data from 10 non stone forming people who had surgery for other 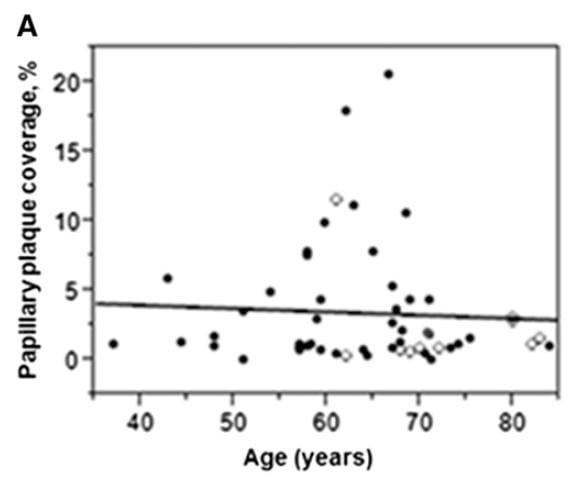 reasons. It is a very valuable paper because it helps to clarify some of the differences between the two groups of researchers.
reasons. It is a very valuable paper because it helps to clarify some of the differences between the two groups of researchers.
Here is panel A from a three panel figure from the paper; the other two panels are simply bar graph summaries of the full data shown on this graph.
A papillary coverage of 5% was used to divide ‘high’ from ‘low’ plaque coverage. This 5% divider was the 90th percentile for the 10 normal values shown as open symbols on the graph.
Although the arithmetic is no doubt true, one notices that 7 of the 10 normals all had plaque abundances at about 2% or less, two at about 2.5% and one high case at about 12%.
This sums up all of the problems in deciding about thresholds to use in calling things high or normal. With one exception the non stone formers have almost no plaque compared to those with stones.
Using the 5% criterion, which is certainly above all but one normal, they found 10 stone formers with ‘high’ plaque and the rest are called ‘low plaque’. 6/10 high and 8/32 low plaque patients were male, and urine calcium was 291 vs. 187 mg/day in high vs. low. So, among their cases they found what we found: men, mainly, with hypercalciuria and abundant plaque, but also what we did not find: Many patients with plaque scarcely above normal, and without marked hypercalciuria.
Moreover, plugging of tubules was found in 3 of the 7 high plaque ICSF who had papillary biopsy histopathology performed vs. 60% (12) of the 20 low plaque ICSF so studied.
Where We Are Now
Both series agree on a kind of ICSF: more male than female, tending to high urine calcium losses ( 291 Mayo, 237 here), plaque surface coverage of 10.5% Mayo, 8% here. But 3 of 7 of these high plaque IH at Mayo had plugging whereas none of our ICSF had plugging despite using similar staining and techniques. So I am forced to conclude that the phenotype of ICSF has in it two distinct groups: Those with considerable plaque alone, and those with plaque and plugging. Because this is a distinction of kind – no plugs vs. some plugs – I am not overly disturbed at the dichotomy but certainly do wonder at why it is.
The ‘low’ plaque abundance of the second group may be poorly phrased in that a glance at the graph of plaque vs. age shows that almost all non stone formers lie in the absolute bottom of the pile so even low – less than 5% surface coverage – is high compared to them.
Furthermore, there is no demarcator: Plaque abundance is a smooth distribution with a high right tail. If asked to describe the data themselves I would say some ICSF have truly low plaque abundance – on line with non stone formers, the rest form a smooth distribution above the non stone formers at the high end of which are – perhaps – the most hypercalciuric patients. I would love to see a graph of plug surface coverage against plaque coverage but I do not have the dataset and cannot draw it.
What to Do?
It is at this kind of juncture one offers – to oneself if to no one else – some speculation or vision of cause, usually phrases preceded by ‘perhaps’.
Plugging might be a later stage of ICSF, for example, high plaque alone, a more pristine picture, the initial phase.
Men and women may differ; there are more women in the low plaque groups, and this may be a clue.
Possibly there are really three ICSF – those with plaque abundances that are as low as the non stone formers, and those with a distribution of higher plaque but with plugs or not.
Time will not resolve this problem, but scientists, measuring, will eventually do so. It is, in the meantime, essential we do not let ourselves be overcome with diversions and try to somehow resolve matters by a resort to models of stones in animals, or in the ways that cells react to crystals. We need to clarify the human phenotype which means more measurements in humans, with the aim of sorting out what is a mixed and complicated assortment of patients.
While we wait, we can say this much to physicians and patients. The trials for stone prevention with water, thiazide, and potassium citrate used ICSF about whom no one knew anything about plaque abundance or plugging so certainly all were tossed into one bucket in each trial. Therefore ICSF who have not been studied with these new refined techniques, and even those who have been, can be treated in the light of the trials, which is all that matters right now.

Hi Dr. Coe, I can’t thank you enough! Boy, do I see myself in this article. (BTW-My last were 20% HA, so I’m just inside that segment.)
This article seems to help begin to address one of my biggest presently unanswerable questions – Why did stones suddenly become a problem around 50 when I’ve apparently had extreme IH all my life?
No surprise, I’m pondering others as well. E.g. what I might be able to do to lower my risk, or at least help keep calcifications from getting worse? IP-6/Phytate is the only lead I have found so far, but it seems like an interesting one. (E.g. http://www.ncbi.nlm.nih.gov/pubmed/10625946 or full text: http://ibdigital.uib.cat/greenstone/collect/laboratoriLitiasiRenal/index/assoc/D123.dir/123.pdf).
Another is whether laser surgery to remove attached stones could have either hurt or helped risk of more stones forming at the same location.
Making progress, slowly but surely, with lots of good help from you and others.
Best regards, Al
Hi Al, Phytate is a theoretical treatment, no real trials, and the author is an enthusiast. Lowering supersaturation with fluids, low sodium intake for hypercalciuria and a thiazide if needed will indeed work, and I hope you do that. Here is a lead article on how to build a prevention model for yourself. Fred
Thank you Dr. Coe. Not to worry, my doctors and I are methodically following the time-tested strategy in that helpful article. That said, I have been looking in parallel for additional ways to specifically address Randall’s plaque. But now I see your point that there is really no need to separate the two because the very same strategy proven to prevent stones should work for plaque too.
Best regards, Al
Hi Dr. Coe
I have hydronephrosis in my right kidney and a parapelvic cyst. Stones form in a pocket in the right kidney and they have been analyzed as calcium based. The largest kidney has been measured at My urologist has recommended both chlorthalidone and urocit K 15. My insurance does not cover the urocit K15. Is there any substitute that I can suggest to him which may be cheaper?
Thank you for your time and assistance.
Hi Anthony, Before the search for a less expensive medication, is your physician sure about the direction of things. If all of your stones are in a cyst or diverticulum, meds will not help – it is a surgical issue. When you say hydronephrosis, if you mean the whole kidney drains poorly, that too raises some concerns. Using potassium citrate will raise urine pH and with inadequate drainage calcium phosphate stones may form. There are a lot of ‘ifs’ here, so I cannot really be sure what to say. If indeed the potassium citrate is right, take a look at the older comments to this article that have lots of purchasing suggestions. Regards, Fred Coe
This is such a wonderful resource for both physicians who treat stones and their patients. Thank you Dr Coe for your time, effort and energy putting this together. I have read many of the papers you have published since I am a urologist, but reading your posts is a masterclass in stone management.
Recently I had a patient in her 50’s who had experienced stone attacks for 20 years, her daughter and aunt have stones as well and they have all been calcium oxalate fitting the profile of idiopathic calcium oxalate stone former. The 24 hr urine had hyperoxaluria of approximately 55, but serum oxalate was low normal. Since her daughter had Ehlers-Danlos disorder a geneticist ordered testing. The geneticist went ahead and since the testing was being done at Yale also ticked the box for kidney stones (Yale has one of the major genetics testing lab in the Northeast). The AGXT region was fully sequenced.
The final report indicated that there was a novel mutation (unreported) in a highly conserved portion of the gene. The Yale lab is now sequencing the aunt and daughter to look for the same mutation. If those come back positive they will sequence the other family members to make sure it is not found in those members. It seems that while polygenetic mutations may be the norm for many stone formers individual families may continue to surprise us with mutations whose phenotypic expression is not as dramatic as a primary hyperoxaluric, but resemble frequent stone formers.
Dear Dr Berry, Thank you so much for your very interesting case. I see occasional cases like this, but have never had sequencing done. Maybe we are all missing opportunities to uncover specific gene variations such as this one. I also thank you for your kind words about the site. Warm Regards, Fred
I am wondering about kidney cysts with milk of calcium found on my ultrasound . Are these calcium stones? My doctor says I have stones based on this. She won’t do urine or blood tests or refer me to a specialist. She says there is nothing to be done unless I pass a painful stone, and that cysts are common (age 64). My urine at times is full of white flecks and I do get abdominal and lower back discomfort at times. Are cysts with milk of calcium considered calcium stones? Should I insist on further tests and a prevention plan?
Hi Deborah, At so great a distance I have minimal insight into the details, but given there is a considerable amount of calcification occurring I would not hesitate to obtain 24 hour and serum testing. Perhaps you do have abnormalities that foster crystallization beside the presence of cysts. So, yes: I would opt to find out why the crystals are forming and whether prevention of more is practical. As for the calcifications, are they all limited to cysts? I would have thought a non infused CT would be of help to be sure that there are not also calcifications elsewhere. I am sure your physicians can arrange for a second opinion from a source convenient to you if you wish that. Regards, Fred Coe
Thanks for some further guidance here for me to pursue.
Thank you Dr. Coe. I always read your articles and admire your work. I am a Pathologist doing Stone analysis at my referral lab. I have been always asked and argued by Urologists that while most of stones are COM and COD in analysis reports, the treatment of all remains same that is plenty of water, calcium, citrate and thiazides , then what is need of getting stone analysis ? Whats could be most satisfactory answer for that ? Please solve my dilemma. I hope you got my point.
Hi Dr Mehta, The problem is that some stones are not COM or COD. Uric acid mixtures, struvite, and the whole treatment changes. Increasing stone hydroxyapatite and there is risk of tubule plugging; urine pH maybe should not be increased with citrate salts – no trial data. Stones of 100% HA maybe RTA. Brushite, very hard, may not be ideal for SWL. Tubule plugs are large and damaging. Stones convert from COM COD to high phosphate, uric acid, and of course struvite. How can you prevent what you do not know? I need to make my article more forceful. Warm regards, Fred
Hi Dr. Coe,
Thank you for all you do. I just read in Arthritis Today about calcium supplement risks. I took Jill’s course and have read your articles, so I know the value of calcium to help decrease my risk of stones. I presently have a calcium oxalate stone in my one and only kidney. I had sepsis in July when the stone blocked my kidney. Now I have been put on a lactose free diet because of diarrhea issues following sepsis. So milk, cheese, etc. are out of my diet for now. I don’t tolerate the lactose-free products, either. I asked my PC what he thought about taking a supplement, and he told me to go for it. Now this article states that “Calcium supplements may also increase the risk of a kidney stone, especially if you’ve had one before.” (May-June 2017 issue). What is your take on supplements as my calcium resources are limited (I do shrimp, broccoli, etc.)?
Hi Jeannette, The diarrhea sounds like c diff from antibiotics and should clear. Calcium supplements are valuable only if taken with meals – in them in that you take the supplement as you eat your food. Use the larger two meals a day. Regards, Fred Coe
I’m 72, have had over 80 cal/ovulate stones. Last month had a dozen from 14mm on down to 9. I’m at wits end. Only pray there’s an answer out here somewhere. Thanks for working on it!
Hi Dotti, With so many stones, have you not been evaluated for cause and treated? The article you commented on has a very reasonable plan for both. Here is another version. I have found that accelerated stones like yours are relatively to prevent, at least in main part. Regards, Fred Coe
Dr. Coe – Thanks for your wonderful work helping all of us who have experienced the pain of kidney stones. In February, I started experiencing chronic diarrhea and a cause has not been determined. I was very surprised by this turn of events because I am a clean eater, exercise, and tend to my health. In July I had my first calcium oxalate stone followed by 5 more calcium oxalate stones in September. Do you believe the chronic diarrhea is related to the onset of kidney stones? Is there a plan of action you would suggest given the additional factor of the diarrhea? Thanks again for all your good work.
Hi Kate, I do believe months of diarrhea can and will cause stones. Your physicians need to cure it. In the meantime maintain your hydration with fluids that contain glucose as they will tend to be absorbed in the upper small bowel. Regards, Fred Coe
Thank you, Dr. Coe. You are the best.
Age 31 with chronic kidney stones currently occuring once to twice weekly. Patient has been diagnosed with Chronic Reoccuring Epstein Barr disease. How does one deal with stones of this frequency. Currently has a stent after having all stones “blasted” in early October.
Hi Lori, I have seen many accelerated stone formers like you describe. Urine risk factors usually look very bland. I lower urine supersaturations markedly and stones generally cease or much reduce in frequency. Do we know what the stones are made of? So we have 24 hour urine and serum testing?? Regards, Fred Coe
Thank you for your great work making this information accessible. I have apparently a rare condition in which the kidney stones in my ureter cause no pain at all. The result was that stones were stuck in the ureter for perhaps several years, perhaps even continuing to grow in that location. When finally discovered, one stone was deeply embedded in the swollen tissue of the ureter. So far, two laser lithotripsy procedures using the flexible uretoscope, each lasting 3 1/2 hours, have removed slightly more than half of one of the large stones that was deeply embedded, and I am scheduled for more procedures to continue, ah, chipping away at the problem.
I have two questions. First, is there a name for my condition, where the ureter does not deliver a pain signal?
Second, would I be a useful subject for someone studying such things, since this is clearly an abnormality?
Hi Michael, Diabetes regularly does this. Otherwise, you have some defects either involving the vagus nerve on in the brain, perhaps hereditary. I could not find any specific causes even in this large review of genetic pain variants. We do not study this problem. I would be very vigilant about painless obstruction – perhaps quarterly ultrasound studies would be a good bet. Regards, Fred Coe
Hi Dr.Coe, Do we know the epidemiology (incidence, prevalence or frequency) of ICSF in the community? I found NHANES data stating about 10% incidence in the US for kidney stones in general, of which 80-90% are calcium containing. But Im looking for a reference for specifically ICSF.
Thanks!
Hi Dr Run, I do not know of population studies that do this. It requires counting people with stones and then filtering out all the systemic diseases. Only clinics do this, and they are not general populations. Best, Fred
The end of the article really hit me…
“Time will not resolve this problem, but scientists, measuring, will eventually do so. It is, in the meantime, essential we do not let ourselves be overcome with diversions and try to somehow resolve matters by a resort to models of stones in animals, or in the ways that cells react to crystals. We need to clarify the human phenotype which means more measurements in humans, with the aim of sorting out what is a mixed and complicated assortment of patients.”
I gave up on potassium citrate helping me because even with taking it I still get multiple stones a month… I have started researching this topic heavily and have seen a small study of 17 people who took N-Acetyl L-Cysteine and Vitamin E. This was done based on rat models and looks promising. I have also seen studies only performed with Vitamin E by itself which again looks promising and also Magnesium by itself which my nephrologist was unaware of, but I guess it’s been in use since the 1950’s?… For patients who have lost hope in conventional treatment modalities, what other options do we have besides experimentation? More potassium citrate?
Don’t take anything I say as medical advice… We don’t know the long term effects of NAC + Vitamin E supplementation I’m just doing it because I have nothing to lose except the kidney stones! I’m a patient not a doctor!
Hi Nick, the article points out that rational planning has reasonable outcomes, so your best bet is a full evaluation with 24 hour urines and bloods to pinpoint the cause, and treat whatever that is with diet or perhaps meds until the cause has been reduced or eliminated. The few trials are good, and the article summarizes them all. Regards, Fred