 WHAT WE DO NOT KNOW ABOUT KIDNEY STONES
WHAT WE DO NOT KNOW ABOUT KIDNEY STONES
Kidney stone matrix proteins glue kidney stone crystals together to make the stones we encounter and treat. Some of the matrix proteins control whether kidney stone crystals form at all, and the rates that they can grow at. Some call these ‘inhibitors’ or ‘promoters of kidney stones, although their exact role in kidney stone formation remains uncertain to today. Many matrix proteins belong to the immune system, or the inflammatory response.
Our Lack of Knowledge
When I joined this field in 1969 we already knew that stones contained an obvious organic component. In my aging and well thumbed ‘Proceedings of the Renal Stone Symposium’ held at Leeds, April 1968 (J A Churchill Ltd, publishers, Standard Book Number 7000 1421 7) Professor WIlliam Boyce wrote a wonderful review of stone architecture that focused on matrix. In it, he set the case forward in a ringing affirmation: ‘The best reason to consider calculous matrix is because it is there. In every human urinary concretion the matrix is present from centre to surface.’
Limitations of Analytical Methods
The sheer analytics styme us. No techniques until now have permitted resolution of matrix into its molecular components. In 2014 I reviewed what I found concerning the proteome of stones, and that review lies below the present lead article by Witzmann. His work uses the most modern techniques and is a great advance over what has been.
Witzmann et al.
The Main Results
Using the methods detailed in the paper, we identified a total of 1,059 unique proteins in two human calcium oxalate kidney stones.
Sheer prolixity, for want of a better word, of nature makes it hard to understand what these proteins do. Probably many are irrelevant to stone formation and simply ride along with the crystals or even other proteins doing no useful work at all. But many are calcium binding and therefore could alter crystal dynamics.
As a group the proteins in these two stones are known to have roles in the immune response system, inflammation, injury and tissue repair which raises the question of whether or not stones cause injury or inflammation or simply that these proteins, fitted to respond to invaders whose walls are generally highly charged are themselves anionic and prone to attach to crystals.
If you want to peruse the entirety here is our spreadsheet. There is also one with the paper itself.
Comments By Frank WItzmann
A New Method
Because our label-free quantitative mass spectrometry (LFQMS) platform has performed well in the past, we used it to extract protein from kidney stone powder and identify and quantify as many proteins as possible. Since previous attempts seemed inadequate for technical reasons, we thought our more comprehensive analysis might give us unique insights into stone formation.
We have quantified 1,059 unique proteins in the organic matrices of two CaOx stones. They corroborate and expand on previous observations of the kidney stone matrix protein composition. As well, they reveal a more complex matrix proteome than previously reported for human kidney stones of any type. But do we have new testable hypotheses concerning how the matrix predisposes to stones?
Does it Matter?
The simple comparison of two CaOx stones may not be enough to answer that question. The two differ markedly. Only one contains neutrophil-associated proteins like calgranulin C, azurocidin, cathelicidin antimicrobial peptide, defensin 4, beta-defensin 1, neutrophil elastase, gelatinase-associated lipocalin, and lysozyme C. Only the other contains kidney proteins like biglycan, dermicidin, destrin, napsin-A, osteoclast-stimulating factor 1, tight junction protein ZO-2, and uroplakin-3a.
Do proteins like these point to unique stone formation mechanisms?
Despite the limitations of our initial proteomic analysis, we believe our approach will enable us to analyse and compare comprehensive protein profiles in small stone specimens from individual patients. We have already studies kidney stone samples from male and female cohorts with CaOx and CaP (brushite) stones. We will share our conclusions here after the CaOx – CaP comparison is published.
The papers below are ones I reviewed over a year ago and remain of interest.
Canales et al
Canales a nd his colleagues extracted the organic material from seven pure COM stones and found 68 proteins. Of these the authors considered as previously unidentified in stone matrix. Myeloperoxidase chain A (MPO-A), α defensin, and the the calgranulins (there are 3 found so far in stones) may be part of the immediate immune response. In addition they found three cell injury proteins, one stress response protein, an array of plasma proteins such as albumin, hemoglobin and transferrin, carbonic anhydrase, and a mixture of proteins that include osteopontin. Finally they found cell adhesion, membrane transport, biogenesis, cell signalling and coagulation proteins. They found Tamm Horsfall protein as most of us would expect.
nd his colleagues extracted the organic material from seven pure COM stones and found 68 proteins. Of these the authors considered as previously unidentified in stone matrix. Myeloperoxidase chain A (MPO-A), α defensin, and the the calgranulins (there are 3 found so far in stones) may be part of the immediate immune response. In addition they found three cell injury proteins, one stress response protein, an array of plasma proteins such as albumin, hemoglobin and transferrin, carbonic anhydrase, and a mixture of proteins that include osteopontin. Finally they found cell adhesion, membrane transport, biogenesis, cell signalling and coagulation proteins. They found Tamm Horsfall protein as most of us would expect.
Merchant et al
Merchant and colleagues did much the same kind of work. From stones of 5 people not otherwise described they identified 58 proteins, of which 11 were high abundance and 10 of these present in all three analyses of the extracts from stones. Of these, all but 1 had been previously been identified as binding to CaOx or HA surfaces. Four proteins were prevalent and high abundance on the basis of normalized abundance computation: calgranulins A and B, apolipoprotein A-1, and THP. Using ingenuity analysis, 3 principle pathways were identified: tumorigenesis, immune, and inflammation. The top canonical pathway was acute phase response signaling. The serious weakness of this work is in the stones: unknown crystals.
did much the same kind of work. From stones of 5 people not otherwise described they identified 58 proteins, of which 11 were high abundance and 10 of these present in all three analyses of the extracts from stones. Of these, all but 1 had been previously been identified as binding to CaOx or HA surfaces. Four proteins were prevalent and high abundance on the basis of normalized abundance computation: calgranulins A and B, apolipoprotein A-1, and THP. Using ingenuity analysis, 3 principle pathways were identified: tumorigenesis, immune, and inflammation. The top canonical pathway was acute phase response signaling. The serious weakness of this work is in the stones: unknown crystals.
Okumura et al
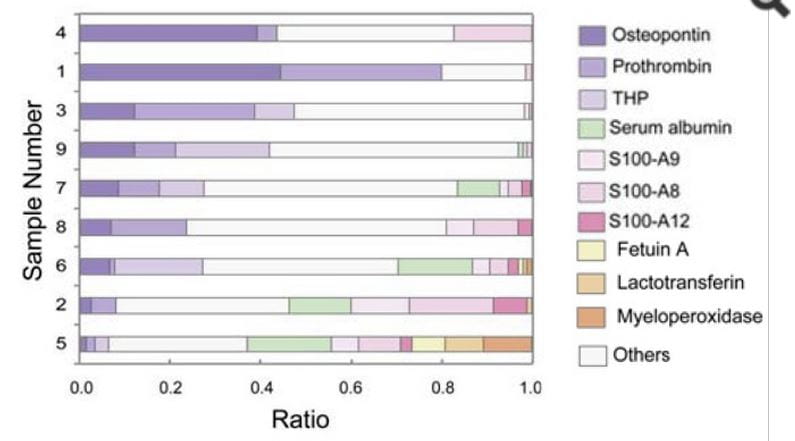
Okumura et al obtained stones from 9 patients otherwise unidentified. All were predominant CaOx by FTIR. Because they found very heterogeneous electrophoretic mobilities of certain matrix proteins, such as THP, they decided to do in solution protease digestion followed by proteomic analysis. This differs from most other studies which did peptide digestion and analysis from bands of gels.
All of the stones h ad some osteopontin, prothrombin F1 fragments, THP, calgranulins A, B and C, myeloperoxidase, and albumin (See Featured figure at the top of this post). What is striking is the variation of protein abundances among the stones. The PTF1 fragment abundance varied over 10 fold; for THP variations were slighter. Calgranulins A and B were almost inverse to PTF1 but ran together.
ad some osteopontin, prothrombin F1 fragments, THP, calgranulins A, B and C, myeloperoxidase, and albumin (See Featured figure at the top of this post). What is striking is the variation of protein abundances among the stones. The PTF1 fragment abundance varied over 10 fold; for THP variations were slighter. Calgranulins A and B were almost inverse to PTF1 but ran together.
This variability itself suggests profitable research. Some of the variability surely arises from heredity. It may cause part of the well known heritability of stones themselves. Possibly variability reflects the effects of stones or of the tissue injuries from procedures used to treat stones.
More
I redacted this article from a longer review you can reach through this link. Although I made no effort to pretty it up, you may enjoy it. If I have missed important references, perhaps yours, please let me know.
Some questions
We know more and more about the molecules. But we have almost no knowledge about how it all works. Are these stone matrix molecules simply surface active components of urine that adsorb to crystals as they form? Do they affect how crystals form, and become part of stones? Do they bind crystals together to make clinically important stone mass? Being in immune and inflammatory pathways, are some of the molecules a response to stones: does having stones set the stage for more stones by altering the urine proteome?
We have far better tools than in 1969. Do we have new testable hypotheses concerning how the matrix predisposes to stones?

what is the best method to remove kidney stone?
There is no best method, because best is related to the kind of stone and the person being treated. Perhaps one might say a single stone in the renal pelvis might be ideal for shock wave lithotripsy, but even here: How big is it? Is there a chance it is uric acid? Are there any complicating issues that make shock waves undesirable. Would flexible ureteroscopy with laser disruption be better for any reason? Surgeons exert considerable judgement in choosing how to remove a stone and I would always be guided by that judgment. When two approaches are deemed more or less the same, patients can choose. Regards, Fred Coe
6 weeks ago I had scope with lithotripsy done, 4 weeks later pain arrived and I was back in ER. 2 weeks after I had another lithotripsy. Is this common for reoccurring stone patients? And can lithotripsies cause damage when having done so often?
Hi Dawn and thanks for the interesting question. If by lithotripsy you mean external shock wave lithotripsy 3 in 6 weeks seems frequent enough you would certainly want to stop these stones from forming. The first event however sounds like endoscopy with laser lithotripsy, which is altogether different. In general, modern urologists are very well informed about the risks and benefits of their surgical modalities and the AUA has published some excellent guidelines for them to assist in thinking about which ones are best. Your problem sounds like frequent stones that need prevention treatment, so procedures will stop being an issue. I would certainly focus my attention on prevention, and hope you can obtain it as soon as possible. As for shock wave lithotripsy and renal damage, it is not a dramatic risk thus far in your case, at least that is how things seem from a distance, but I am sure you and your doctors will be a lot happier when stone recurrence is ended by effective medical prevention. Regards, Fred Coe
Thank you for your reply Dr Coe. I have been battling these stones for almost 20years, but seems for frequent the last few years. Dr Patel has referred me to you. As soon as I complete my tests, I will be making the appointment. I look forward to the prevention process. Light at the end of the tunnel.
Again, thank you for your time in replying.
Dear Marie, I am happy to be of help. Since Dr. Patel referred you here, I will be pleased to see you and do what I can for stone prevention. Fred Coe
HI Dr. Coe,
I’m gathering all of my info, so for my next appointment I can be more prepared. Right now I’m doing my 24 hour urine test, just got my blood work done which came back normal and got a KUB. I had a CT scan back in Nov. 2016, and I have 26 STONES!!! I’m only 39 years old and a female. Three years ago was the first time I discovered I had kidney stones. 17 in my left kidney and 9 in my right, right now. I asked what the stones are called and they’re called Carbonite Apatite stones. Is there another name for my kidney stones??? I can’t find a lot of info on my stones on the Internet. I take Urocit K but not all of the time because it gives me heartburn. Is there anything you recommend I ask in my next appointment? I’m really stressed with the amount of stones I have and just don’t want to keep going through this if I’m doing something myself to cause these. Thank you for your time. P.S. not sure if it matters, but I had four kids in the past eight years. My last baby exactly a year ago.
Hi Mandy, YOur stone is calcium phosphate. Here is an article about stone types. Here is one about your type of patient. Here is a article about how someone like you needs to be treated. Here is an article about how to prepare for your visit. Finally, here is one about the steps to stone prevention. Now, you are well equipped with information, and it is reliable. Good luck, Fred Coe
Thank you so much for the information. I really appreciate it!
I have been passing little red crystals in the urine, I feel them in the uriter when passing. I had them sent to a lab at N.S Manhassett Hospital. They came back as not stones, but rather protein blood mix. What are they From? And is there anything I can do to prevent them in the future?
Thank you
Hi Steven, These may indeed be blood clots. You need evaluation for this by a urologist who can find the source of the blood and fix things for you. Prevention is to find the cause and get rid of it. Do not omit to get a full evaluation for this from your physicians. Regards, Fred Coe
Dr Coe, I have MSK and have had many episodes with kidney stones since age 15. I am 68 now. I need spinal fusion for scoliosis and have been taking Forteo for 3 1/2 mos. Anout 2 wks ago I began having flank pain and as of an ultrasound two days ago, I have clusters of new stones in both kineys. My spine surgery requires my bone density to be improved to assure fusion. Is there anything I can do to prevent making more stones in the future, and should I d/c the Forteo? Thanks for any advice you can provide.
Hi Rae, Forteo is parathyroid hormone that can increase urine calcium and foster stones. You need it for your bones. Very low sodium diet would help lower urine calcium, and if combined with high fluids might stave off more. Ask your treating physician. Finally a low dose of thiazide – like agents like chlorthalidone 12.5 mg every other day – if combined with low sodium diet could lower urine sodium even more. Regards, Fred Coe
My 79 year old mother has passed several large Matrix kidney zones. I had her save the stones to show the specialist. He said he had never seen anything like it in his 27 years of practice. He sent them off to be test so we now know they are Matrix. Mom had Scan with contrast for bladder and kidney and two stones were sited; one in both ureters. Scoping procedure to the bladder was done as it was completely clear. Due to the radiology report findings of Matrix and blood, the doc wants to scope to the kidney to see what is causing these stones. From your research and experience, will you speculate what they could possibly find? I’m not sure it is absolutely necessary at the his time. Please advise ASAP.
Thank you in advance,
Kimberly
Hi Kimberly, You are the second person this month reporting matrix stones. Radiology cannot diagnose such stones, only stone analysis, so I presume stones have been so analysed. If not, if this is just radiology, I would not rely on it. If you have the stones, get them analysed – they may contain some crystals that are organizing the protein into these stones. Regards, Fred Coe
Hi there, I’m interested in knowing more about kidney matrix stones. I’ve recently passed a few “soft” stones that were sent for calculi analysis and came back as trace Calcium Oxalate and protein (protein type not specified). The stones were mailable, yellow (some with darker red spots) and felt almost rubberized. Following passing these stones I had renal colic, flank back pain and began having symptoms of a UTI. Shortly after I was tested for infection which came back positive. I was treated for infection with oral antibiotics (nitrofurantoin). A few days after treatment my pain increased and went to ED due to high pulse and high blood pressure. Once treated with IV antibiotic I began to improve. Half a year before this I almost lost my life to sepsis after passing an infected stone and so those symptoms were alarming. A few months ago I had a 13mm stone removed from my left kidney that was composed of several stones “glued” together with infection. My right kidney has a 6mm stone which is believed to be infected, since I’ve been having repeated UTI’s and pain. I’m scheduled to have the stone removed but the date of surgery keeps getting pushed. Is what I am describing sound like the symptoms and physical components of a matrix stone? Or could this be another type? I have a history of calcium phosphate stones, elevated PTH (negative sestimibi), vitamin D hydroxy 25 deficiency (47 nmol), sepsis from infected stones and frequent UTI’s. Unfortunately our community is undergoing a health care crisis related to urology and I don’t have access to specialists. Any advice would be greatly appreciated.
Hi KC, Very complex. Infection seems your dominant issue. I am guessing sepsis has been with Proteus, Klebsiella, Pseudomonas, or Enterobacter species, and that your stones may have struvite in them. That your community is having a health care crisis worries me. Can you possibly afford to travel to a medical school that has a kidney stone program? I would advise it given what sounds like complex problems and a lack of available physician manpower. You can look on the web for places most convenient for you. I am concerned you get care sooner than later. Regards, Fred Coe
Thank you for your concern- it is unsettling that we are being faced with this in our community and it’s is unfortunate that it is due to large volume of cancer patients/lack of doctors. I am Canadian and am considering travelling over the border for care (we are a border town with UP Michigan). I still do not have a date booked for surgery, but I’ve been told at some point by the end summer/early fall. I feel that I will end up in ED before the ureteroscopy since my pain level continues to be high. I’ve been given narcotics to deal with the pain but I often refuse to take it unless I cannot function- to me, it is not a solution that I want to accept. My urologist is wonderful- he is just swamped. The only option to get surgery sooner would be to be in critical condition in ED and be put on a “wait and see list”. That being said, do you know any specialists in the Michigan area? We also have family in Chicago. I’m a mother of two young babies so we want to limit travelling long distances. I’ve also kept one of the soft stones in case I can send it to a better testing facility but the stone has decomposed quite a bit (given, it is made of protein, I am not surprised). Is there an institute I can send this stone to? Thank you again for your reply,
Best,
Hi KC, Litholink does stone analysis but you need a physician order and I do not think they serve people in other countries – like Canada. In Michigan is Ann Arbor which is very well reputed. In Chicago we have many institutions including my own, but remember there is an insurance and payment issue in US. Regards, Fred Coe
Hi Dr.Coe:
I have been passing what my doctor thinks is Matrix stones for 12 months now, and my symptoms are getting increasingly worse each day. Is there a specific analysis that can be done on the soft stones? They have done many CT’s and MRI and ultrasounds and each have shown nothing. I have had reacquiring utis that have not been treated because my family doctor saw the growth but the bacteria was mixed each time. So they wouldn’t treat the infection even though I had high white blood cell count for months.
Is there anything you can suggest test wise that I can ask for, and what can be done to prevent more from forming? I’m desperate
Thank you in advance!
Hi Kristy, Possibly the matrix stones are forming around crystals and FTIR stone analysis will reveal them. This means sending samples for stone analysis. Litholink (I have no financial relationship) has outstanding stone analysis as does Mayo Clinic. Possibly infection is causing the problem and the bacteria evade common cultures. Perhaps your physicians might want to order more sensitive testing for this. Both special media for culture and gene testing are now used to identify uncommon infections. Regards, Fred Coe