 My book chapter makes the prime point that low urine pH causes uric acid to crystallize in urine and produce kidney stones. But it does not detail what lowers urine pH to such an extreme. In part, bowel diseases lower urine pH. That is another subject altogether, because they also cause calcium stones. So they need their own articles. But in the main, low pH arises in people who are obese, have metabolic syndrome, diabetes, vascular disease all in some combinations. As an alternative,sometimes low pH associates with gout and none of these are present. In either case, this article concerns what kidneys do to lower pH in the absence of an obvious systemic cause.
My book chapter makes the prime point that low urine pH causes uric acid to crystallize in urine and produce kidney stones. But it does not detail what lowers urine pH to such an extreme. In part, bowel diseases lower urine pH. That is another subject altogether, because they also cause calcium stones. So they need their own articles. But in the main, low pH arises in people who are obese, have metabolic syndrome, diabetes, vascular disease all in some combinations. As an alternative,sometimes low pH associates with gout and none of these are present. In either case, this article concerns what kidneys do to lower pH in the absence of an obvious systemic cause.
This article parses out and pulls together the work of the Dallas group that has contributed most of what we know about low pH in uric acid stone formers without bowel diseases or other obvious causes. They have presented their work in many review articles, so this one should be taken as a public tribute to them rather than an academic summary. Even so, it is fearsome technically and not for everyone to undertake. A principal author of the Dallas research, Dr Khashayar Sakhaee was kind enough to read the final manuscript and found no errors.
As as personal mote, I put here some of my own data collected over decades with Joan Parks, my long term colleague. Though merely clinical, it confirms some of what the Dallas group has found, though it cannot reach the depths of their science.
The drawing by Ramelli (Agostino Ramelli’s Le diverse et artificiose machine Paris, 1588) shows a machine for lifting things. The mechanisms depicted in this 16 century drawing reflect our interest here in how things work. “Ramelli was born in northern Italy, probably in 1531. As a young man he served under the famous Italian warlord, Gian Giacomo de’ Medici, Marquis of Marignano, and became trained in mathematics and military engineering. His reputation grew and he eventually left for France to serve under the Duke of Anjou, later King Henry III. ” – From notes at link above.
Obesity
Obesity itself can do it.
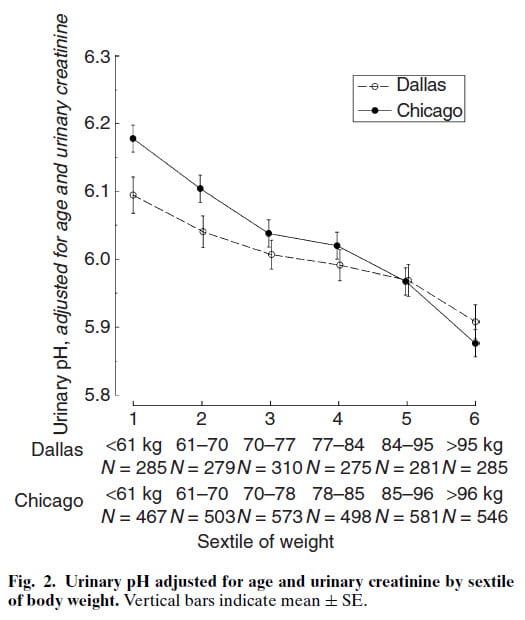
This figure illustrates that effect in urine pH from thousands of kidney stone patients plotted against sextiles of body weight. Adjustment for urine creatinine, a measure of muscle mass, converts weight into a measure better related to BMI. We included age, because urine pH can fall as one grows older. Our own lab in Chicago and that of the kidney stone research group in Dallas collaborated.
No doubt, as weight rises urine pH falls. But how can being fat lower urine pH?
Low Urine Ammonia
Long before the remarkably important work by Khashayar Sakhaee, Orson Moe, and Charles Pak that has more or less given us our modern view of pathogenesis, scientists had blamed low urine ammonia excretion for causing low urine pH. They blamed it for good reasons. Here in the West our diets impose an acid load kidneys must excrete. They excrete that load via ammonia and by titrating urine phosphate. The latter requires urine pH fall. If ammonia production lags, urine pH must fall more for a given acid load.
Note that urine ammonia is mostly in the form of the ammonium ion NH4+. Ammonia itself is in vastly lower concentration. So in this article, we refer to the ion as ‘urine ammonia’ understanding that we refer mainly to the ionic form.
Not Obviously Low
Sakhaee and colleagues performed the first fully controlled test of this idea. They fed subjects a constant very low calcium diet (400 mg) and collected 24 hour urines in the clinical research center on days 6 and 7 of that diet.
They did not find low urine NH4+ by simple measurement. Urine NH4+ excretion was the same in normal people, and those who formed uric acid or calcium oxalate stones – 32, 39, 33 mEq/day. They did find a lower urine pH in the uric acid stone formers, of course: 5.96, 6.02, and 5.40 for normals, CaOx and uric acid stone formers, respectively.
The lower urine pH should have increased urine NH4+, all thing considered. Ammonia is ‘trapped’ in the collecting ducts as NH4+ as pH falls. The lower urine pH would have ‘trapped’ a higher fraction in uric acid stone formers. So their urine NH4+ ‘should’ have been higher given the lower urine pH.
Low In Relation to Urine pH

Urine NH4+ (what is trapped) – vertical axis – should rise as urine pH (horizontal axis) falls. But for uric acid stone formers (black circles), if anything, it fell. Startlingly enough, values at low pH matched those at high pH – when trapping is poor and low NH4+ is expected. As well, points from mixed uric acid/calcium oxalate and uric acid stone formers (black down pointing triangles) matched.
But a few normals (open circles) and calcium oxalate stone formers (unfilled down pointing triangles) had about the same NH4+ excretions at pH around 5.5. This obscures any unique claim for uric acid stone formers.
Even so, one suspects trouble with ammonia production when NH4+ fails to soar upwards as pH falls so low as 5. For example, the reference links to a an old research by Schwartz and Relman. When they lowered urine pH very rapidly in humans without changing blood pH. urine NH4+ rose remarkably. Why not observe that in uric acid stone formers?
Low In Relation to Net Acid Excretion
The total net acid excretion (NAE) was the sum of NH4+ and titratable acid minus bicarbonate and citrate ion. This last is unusual. Most conventional calculations omit the loss of citrate. LIkewise, a remarkable and recent analysis omitted urine citrate. Yet citrate can be metabolized to bicarbonate, so losing it means losing potential base.
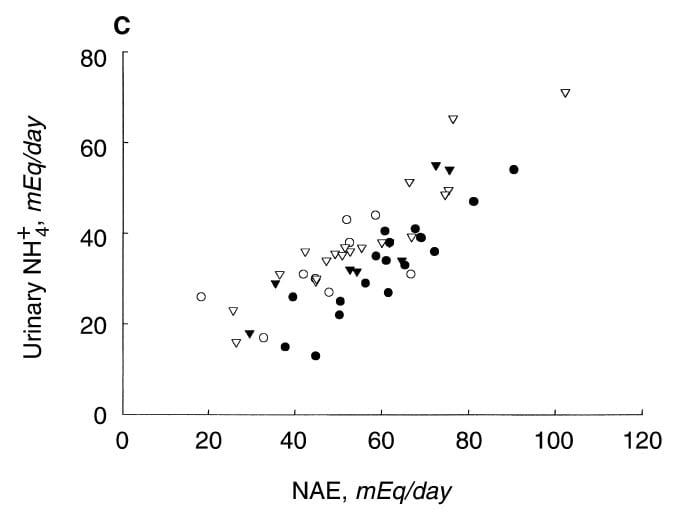
This graph is more compelling. As NAE rises NH4+ rises with it. But the uric acid stone formers trail along the lower border. Compared to the other patients they have less NH4+.
But if NH4+ is so low in relation to NAE, what makes up the difference? What else but titratable acid or less citrate loss? In fact both matter, but TA matters more. Values for uric acid stone formers were 33 mEq/d vs 23 for normals and CaOx patients. CItrate was a lesser player: 4.4 vs. 5.6 uric acid VS, CaOx patients, but normals lost 8 mEq/d.
So, uric acid stone formers mainly lose less NH4+ and make up the loss with a lower pH that titrates urine phosphate with more protons. The net result is about as much NAE – even more – than normals or CaOx stone formers.
But, does all this really prove a ‘defect’ in ammonia production? What if the low pH were a thing of itself? The lower pH would titrate more phosphate, TA would rise, and the demand for ammonium ion to remove acid would fall. However attractive, the work so far is not enough to tell the one from the other.
Inadequate Response to Acid Load
The way around this uncertainty was to do an experiment, not observations. They gave an acid load and asked what happened to urine 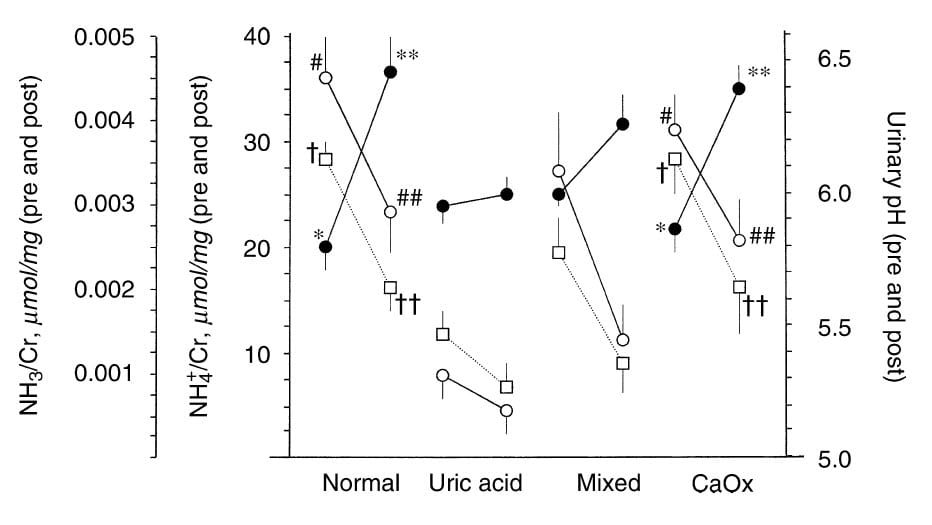 NH4+ and pH. Focus on the uric acid column. Urine pH – squares – fell from low to very low. Urine NH4+ (black circles) hardly rose. Ammonia itself – far left vertical axis – fell because low pH raised the fraction of NH4+.
NH4+ and pH. Focus on the uric acid column. Urine pH – squares – fell from low to very low. Urine NH4+ (black circles) hardly rose. Ammonia itself – far left vertical axis – fell because low pH raised the fraction of NH4+.
Normals and CaOx stone formers raised their NH4+ greatly – look at the change in the black dots. The mixed patients were mixed; their rise in NH4+ was in between.
Good Science
Is this good science? Yes. The idea of some defect in ammonia production led to a crucial prediction. If present, then urine NH4+ would not go up normally with an acid challenge, because limited by production.
Since that was the case, does the outcome prove defective ammonia production?
No. It merely says the idea passed a real test that could have falsified it. For the moment the idea is sound and became the basis for more research.
What Lowers Urine Ammonia?
The idea arose from associations. Gout associates with uric acid stones and also with metabolic syndrome – itself a state of insulin resistance. Insulin stimulates ammonia production, and insulin resistance would therefore reduce ammonia production. Thus the notion arose that insulin resistance caused the low NH4+ in the urine – of uric acid stone formers.
But how to test this? For example, can we find evidence of insulin resistance in uric acid stone formers?
Are Uric Acid Stone Formers Insulin Resistant?
Thirteen such patients compared to 55 people who did not form stones showed higher body weight, blood pressure, BMI, serum cholesterol, LDL cholesterol, and fasting blood glucose. These traits more or less define the metabolic syndrome. But insulin resistance requires a more specific test. You need to infuse insulin and glucose, and measure the rate of glucose you must infuse to keep the blood glucose in the normal range. Insulin resistance lowers the rate of glucose utilization. So less needs be infused per minute. This procedure is called normoglycemic hyperinsulinemic clamping.
Is it Age and Weight or the Low pH?
The thirteen uric acid stone patients used up 6.3 mg/min/kg of lean body mass during the clamping, the normals used 9 (p=0.008). So the patients indeed had insulin resistance. But the patients were a lot older and fatter, leaving open the question of whether insulin resistance was a necessary counterpart of uric acid stones or just age and obesity. A special analysis of 13 normals matched in age and fatness to the 13 uric acid stone formers was not encouraging. The glucose disposal rates were 7.8 and 6.3 (p=0.2). No important difference. But those fat older normals had a uric pH of 6.1. And, even more, their urine NH4+ was higher in relation to net acid excretion compared to the uric acid stone formers.
So, just the fatness and age of the uric acid patients could not account for the low urine pH, nor could insulin resistance per se because in this special 13 to 13 head to head comparison glucose utilizations were no different.
A Brilliant but Incomplete Response
Here, the scientists had reached a wall.
They needed a new path and chose this one. They had collected timed urine collections in 33 of their normal subjects before and during insulin clamping. This let them ask if insulin infusion could raise urine pH and, more importantly, urine NH4+. It could. Urine pH rose from 6.1 to 6.8, NH4+ from 2.7 to 4 mEq/2 hours, and citrate from 48 to 113 mg/2 hours. All are highly significant changes with probabilities from chance below 1 percent.
So insulin could in principle have raised the urine pH and NH4+ and citrate levels for uric acid stone formers. But, alas, they did not have the urine samples from them so we cannot be sure.
Diabetes and Uric Acid Stones
Remarkably able scientists, the Dallas group chose yet another way to specify the defect causing low urine pH in uric acid stone formers. Diabetes and uric acid stone formers share alike a low urine pH, but insulin resistance did not fully explain the low pH in uric acid stone formers. On the other hand, their preliminary results from acid loading had disclosed an obvious defect in the latter. What better than to ask in a head to head experiment if diabetics also had the same defect.
Who They Studied
They gathered 13 patients with type 2 – adult onset – diabetes well matched in age and obesity with 14 non diabetic uric acid stone formers. As controls they used 8 people with no stones or other disease. The uric acid stone formers produced 100% uric acid stones. Final average BMI values matched well – 32 – 34. But the uric acid stone formers were almost all men – 13/14, the diabetics less so 8/13 male.
The diabetics and uric acid stone formers had similar low urine pH values – 5.49 and 5.41, but the normal people were higher 5.92. Both patient groups had low urine NH4+ as a fraction of their net acid excretion compared to the normals – 0.6 06 vs. 0.9. So, they assembled a proper grouping for the test. When challenged with acid, would the diabetics behave as the uric acid stone formers? Or not?
The Test

I show here panel D from a large figure because the one panel gives the key information.
For background, the urine pH fell in all three groups to more or less the same extent. Naturally net acid excretion and titratable acid excretion rose, too.
Here is the NH4+ story. The controls showed a rise – the normal or expected one. The diabetic patients rose, too. This was a key finding. These diabetic people had the same abnormally low urine pH as the uric acid stone formers. Their urine NH4+ excretion was low in relation to their total acid excretion. But, when challenged, they rose normally.
The uric acid stone formers behaved here as in the past. They could not raise their urine NH4+ losses normally. For those with statistical tendencies, the uric acid stone formers were below both the normals and the diabetics, but the diabetics and normals did not differ from each other.
So There You Have It
Uric acid stone formers may resemble diabetics in having a low urine pH. Insulin may have a lot to do with ammonia excretion. Uric acid stone formers may indeed – not proven – have insulin resistance. But, diabetics, who certainly are insulin resistant, do not have the ammonia defect uric acid stone formers have. The two groups differ. Why diabetics have so low a pH remained unsettled at paper’s end. LIkewise, the reason for the ammonia defect in uric acid stone formers.
Is This All?
In a way, yes. The Dallas group has done much more with uric acid stone formation than I have reviewed, but I believe I have presented the most critical findings. I did not make it an issue, but total acid excretion is high among uric acid stone formers for reasons not known. Likewise, although their urine pH is just about as low, diabetics do not all form uric acid stones. But uric acid stone formers were chosen for their stones, so comparison of risk – low pH, vs. outcome – uric acid stones cannot be made.
You might wonder if I have slighted other investigators in favor of the Dallas group. But I have not. Many have helped define the uric acid stone phenotype, and establish the preeminence of low urine pH. I gave them what I could of review in my clinical article. They did not add to what I have presented concerning how the low pH comes to be.
And so, I leave off here and say, simply, the Dallas group has given us what we have of modern pathogenesis of uric acid stones. We should thank them.
Can I Add Anything?
Because Joan Parks left me the marvelous file of data from uric acid, calcium oxalate, and calcium phosphate stone formers I used to illustrate the clinical uric acid stone article, I can add some obvious empirical information. Note that I used log scales for the graphs to provide visual clarity but performed my analyses on untransformed data having no reason to transform them apart from visual appearance.
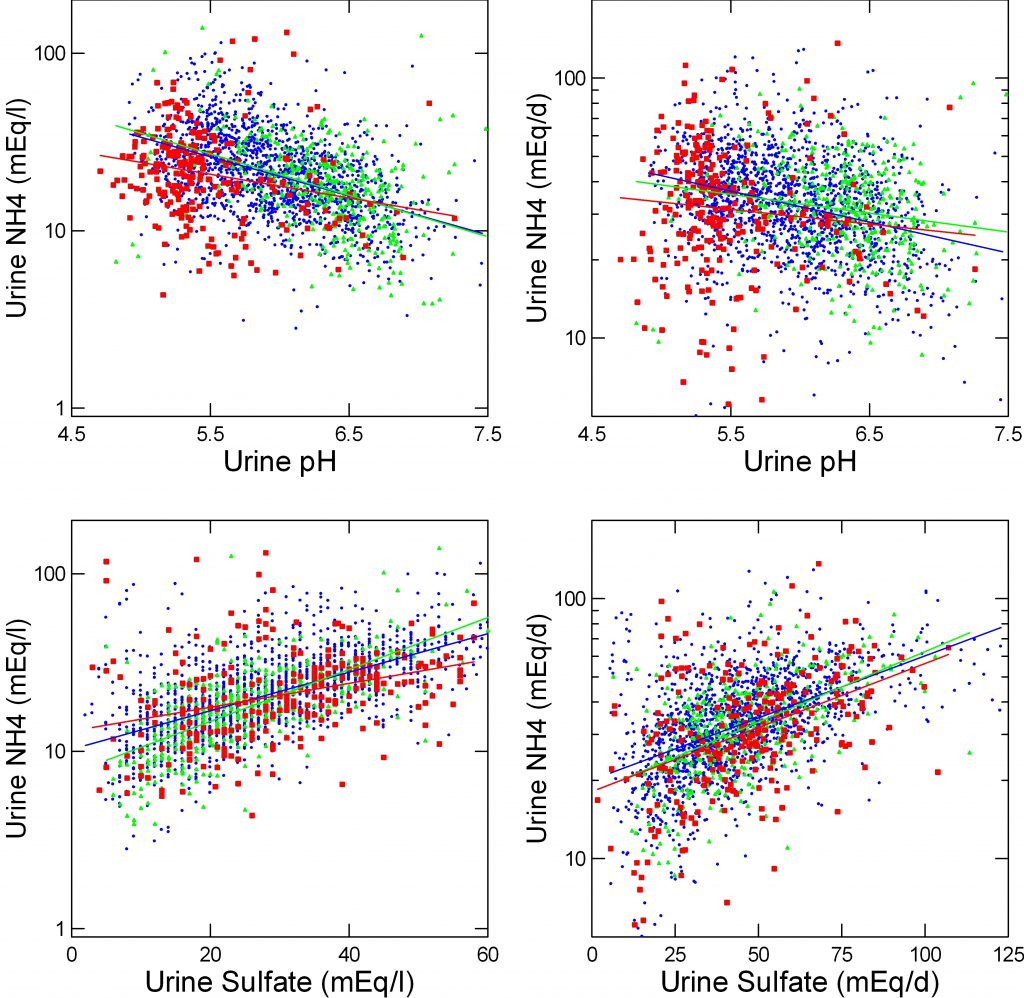
NH4+ Concentration
By pH
As I have mentioned above, urine NH4+ rises as pH falls because the charged ion is ‘trapped’ in the tubule fluid. The upper left panel shows that for uric acid (red), calcium oxalate (blue) and calcium phosphate (green) stone formers.
Because they have a lower urine pH, UA stone formers have a higher urine NH4+ : 25, 24, 21 mEq/l, UA, CaOx, CaP stone formers (CaP vs. UA or CaOx p<0.001).
But, is NH4+ too low when adjusted for urine pH?
No. Adjusted values are all the same – 24, 24, 25 UA CaOx, CaP). pH is the main covariate – F=92, p<0.0001. But the difference of slopes is significant (p<0.00001) and it is the UA slope that lags the rest suggesting a reduced increase of NH4+ with falling pH (per one pH unit, -11/5, -11.2 and -2.8, CaOx, CaP, UA).
By Sulfate and pH
I cannot calculate NAE as the Dallas group did as I have only 24 hour urines and bicarbonate values would be suspect. But urine sulfate strongly approximates net acid load. A plot of urine NH4+ against urine sulfate (lower left) shows a powerful correlation as expected. In a general linear model, sulfate effects dwarf those of pH (F=251 vs. 24). The slopes of urine NH4+ on sulfate are different and that for uric acid is lower. For a one unit change in sulfate the average change in NH4+ is 0.469 mEq/l. But for CaOx SF this is 0.506, for CaP SF 0.719 whereas for UA SF it falls to 0.227 (F=14, p<0.0001 for variation). Once again, NH4+ response lags. Fully adjusted, mean values of urine NH4+ are almost identical. Note that urine sulfate is rounded to the nearest integer giving rise to the vertical pixelation. Also note that the red dots do not run below the blue and green dots: there is no intercept difference as was found when urine NH4+ was plotted against urine net acid excretion by the Dallas group.
NH4+ Excretion
By pH
I would not overly burden this text with a parallel analysis of NH4+ excretion rates except that CaP SF produce higher urine volumes which in turn changes the relationships of NH4+ excretion rates between the groups: Volumes were 2.017 vs. 1.739 and 1.672, CaP vs CaOx and UA SF, respectively, p<0.001 CaP vs. either.
As for NH4+ itself, NH4+24 varied with urine pH (upper right panel). The pH stope dependency differed among the three stone forming groups, and the slope for UA SF was lower than for CaOx SF: -4.671 and -2.272 respectively.
By Sulfate and pH
As for NH4+ itself, NH4+24 varied with urine sulfate F=284, p<0.0001. But slopes were marginally different F=3.87, p=0.021. Fully adjusted urine NH4+24 were the same for all three groups – 36 35 37 mEq/d CaOx, CaP, UA. Values for NH4+24 do not run toward the bottom of the dots, of course, given the adjusted values were not different.
What About Sex?
Sex often matters, so I added sex to each of the regressions using all the proper forms. It has no effects of any kind. I am surprised but one takes what one is given. It alters neither the intercepts nor coefficients to any significant extent.
What Have I Added?
In a crude observational study whose only virtue lies in large numbers of samples (about 2,000), one finds some reduction of ammonia in UA stone formers. This is true for concentration based analysis more than excretion. Slim as it is, my mote supports the idea that something is really wrong with ammonia in UA stone formers compared here not to normals but to CaOx and CaP SF.
Clinicians need not bother to look for low urine ammonia excretion in their UA SF as crude simple lab testing cannot reveal it. It is too pH sensitive, for example. The effect – something wrong with UA SF – certainly exists. I Found traces of it. Perhaps my colleagues in Dallas will one day identify it more completely.
So take this as my homage to my friends in Dallas.


Thank you for your efforts
This work associating UA stone formation with obesity, insulin resistance, and metabolic syndrome is quite interesting and something I have long suspected simply looking at my immediate family’s medical hx. My father,brother,and myself all form (..ed), UA stones and suffer from the constellation of diabetes,obesity, etc.
My first episode with a stone,32y ago…occurred at age 33,when I was quite definately not obese, and not diabetic,and extremely physically active…
Stones have persisted at 2-5 y intervals, obesity now a fact,but not diabetes. Urine ph approx 5.5 without citrate,6.5-7.0 with citrate.
I offer this as one case where the UA stone formation preceeded the development of the other metabolic conditions.
Again,my thanks to you and Jill for what you do.
MB
Hi Nike, So long as the pH stays up you should have no uric acid stones. There are patients without diabetes, obesity and all with low urine pH probably by heredity. Perhaps you are an example. Regards, Fred Coe
Does medications such as Potassium Citrate, loss of weight, and a change of diet necessarily eliminate acid stones, Dr. Coe.
Thank you for this most interesting article. RQ
Hi Ronald, Potassium citrate enough to raise urine pH to above 6 will prevent uric acid stones. Other measures are essentially unnecessary and by themselves futile. Just the one will do all you need. Regards, Fred Coe
Hello Dr. Coe.
Thank you for such a great work on uric acid stones.
I have been suffering from urine uric acid stones and crystals for 6 months now. The family doctor and urologist do not pay attention to the pics I am taking myself from my own urine sediment since I am a research scientist with vast experience in the diagnostic lab. However, after the CT came back normal, the specialist sent me for dynamic tests which also resulted normal. I visited a walk-in clinic and the doctor did a blood uric acid test, which resulted elevated. I am on a diet and 100 mg of allopurinol daily for about 5 weeks now, but not getting completely recovered. I still see the crystals in the urine and I still have burning and urethral pain if I do not empty the bladder regularly.
Your opinion about what is missing will be highly appreciated. Pictures of stones and amazing crystals are available for your evaluation.
Thank you in advance
Adriana Trajtman
Hi Adriana, I gather you have successfully identified uric acid crystals in your urine and have pain and crystal passage. Uric acid crystallizes when its principle hydrophilic site on N9 becomes protonated and the pKa is 5.35 in urine. So the crystallization is stopped by alkali sufficient to raise urine pH to about 6. Usually potassium citrate is used and usually 24 hour urine testing is performed. The latter will disclose a low pH and you can dose to about the mEq of urine NH3 – usually 30 mEq or so per day. Allopurinol is useless. You also mention stones – I presume these have been analysed by FTIR and are indeed uric acid. Regards, Fred
You are a gentleman and a scholar. Your work would seem to overlap with gout. I have no symptoms of kidney stones but do suffer with intermittent bouts of gout. I am a researcher in social sciences so I cannot fully appreciate the biochemistry, but it is clear to me that these uric acid related conditions overlap. Is potassium citrate also you recommend Tx? I would love to see you directly address gout because you seem to possess a vast relevant knowledge. It could be that I lack the technical expertise to connect the dots that are already in plain sight.. Thanks in Advance.
Hi Lyle, The crystal of gout is sodium hydrogen urate, and conditions are set by the blood and joint fluid. So the overall system is like stones but the target is blood urate. Potassium citrate has no role as the gout salt solubility will not be affected. Of interest, many causes of gout involve reduced kidney urate removal and also low urine pH, so uric acid stones and gout coexist frequently. Regards, Fred
Hello Dr. Coe. Do purines in food contribute to uric acid stone formation? Just curious. I have a long gout history, but no uric acid stones. My urine ph is high so no worry there. Just curious to understand if there is a correlation. I was surprised to see it not mentioned.
Hi Chad, NO; it is urine pH, only, for uric acid stones. Fred
Hi Dr. Coe,
I recently had my first stone. The lab analysis of the stone showed 90 percent uric acid and 10 percent calcium oxalate. I am now starting a 24 hour urine test. I have always had a urine pH around 5.5 but never had a stone until recently. However, I did start taking 1mg of prucalopride (Motegrity) about 6 months ago. Do you think the prucalopride which serves as a laxative could have had anything to do with the stone formation?
Thank you so much for your efforts!
Steve
Hi Steve, An increase in colon fluid loss from diarrhea will reduce urine pH and possibly could have pushed things to the point of crystallization. But since uric acid stones are totally preventable with potassium citrate I would just use it. The dose is adjusted upward until the 24 hour urine is about 6 or so. Diet change can help reduce the amount of the supplement but always monitor 24 hour urine to be sure pH remains high enough. Regards, Fred Coe
Hi Fred,
I find potassium citrate upsets my gut and causes me to have a colon cleanse like action . Would baking soda in water be ok and if so what dose?
Hi Ed, if it is for uric acid stones – this article – sodium bicarbonate is fine. I would suggest the same dose, perhaps 40 mEq spread out over 3 or 4 doses per day. There are OTC alkali that would also work. Regards, Fred Coe
Hi Ed, if it is for uric acid stones – this article – sodium bicarbonate is fine. I would suggest the same dose, perhaps 40 mEq spread out over 3 or 4 doses per day. There are OTC alkali that would also work. Regards, Fred Coe
Hi Dr. Coe, medical graduate here! Thanks for the great effort! I really really appreciate your work! I aspire to be a doctor who actually reads & UNDERTANDS medical literature & has a scientific detailed approach to all disease & management aspects!
I’ve uric acid stones but I’ve only managed to get KALICIT effervescent tablets, this prescription’s active ingredients are: 1 tablet contains 2.17 g of potassium citrate, 2.0 g of potassium bicarbonate, 2.057 g of anhydrous citric acid.
Excipients: sucrose and glucose syrup, macrogol 6000, saccharin, lemon flavoring D-Sorbitol.
I’m having real hard time converting this info into practical dosage! I understand that the dose depends on urine pH & then should be adjusted accordingly. I also understood that 10 mEq twice or three or four times is the usual starting dose… What is the equivalent dose if I were to take the aforementioned drug “KALCIT”?
Thanks again doctor Coe!
Hi Baraa, 1080 mg of K citrate crystals is 10 mEq so 2017 mg would be about twice that of 20 mEq of base. The citric acid will not provide alkali as it is metabolized as the acid. The 2000 mg of K bicarbonate will give another 20 mEq so a pill has 40 mEq of alkali which is enough to prevent uric acid stones. I would drink half in the morning and the other half in the evening. REgards, Fred Coe
Thanks for responding Dr Coe! So I ended up doing the exact thing, taking half in the morning & the other half in the evening…. but I’ve been scouring the medical literature to understand this & got contradicting conclusions, does urine alkalization “only” help preventing uric acid stones? or does it ALSO treat them & dissolve them?
Hi Baraa, Uric acid is soluble at pH above 6 to over 1000 mg/l of urate species. Below 5, when it is all protonated it is soluble only to 90 mg/l. So ALSO is true. Fred