WHAT IS POTASSIUM CITRATE
We have reached the point in the evolution of this site where the main stone risk factors are introduced and detailed, and the importance of citrate established. I have written about the price of potassium citrate because many patients and physicians have told me that it has risen steeply in recent months, and I would like to be of help.
A LOUD DISCLAIMER
In this post I will mention beverages and medications by name. Let me be clear: I have no financial relationships with the companies that produce or sell the products I write about here. Likewise neither I nor my colleagues at University of Chicago receive any support, financial or otherwise from these companies.
WHAT DOES CITRATE DO FOR PATIENTS?
It Can Reduce Formation of Uric Acid Stones
Some patients produce too acidic a urine which raises risk of uric acid stones, and they need supplemental alkali to make their urine less acidic. The use of potassium citrate to make urine less acidic will prevent uric acid stones in most patients who form them.
It Can Reduce Calcium Stone Formation in Patients with Low Urine Citrate
Some patients form calcium stones because they produce urine that is low in citrate, a valuable inhibitor of crystal formation. Most of the naturally occurring inhibitors in urine are complex molecules about which we can presently do nothing. But citrate is a small and easily measured molecule which we can prescribe and which will increase the urine citrate in at least some patients. Potassium citrate lowers urine calcium excretion. In so doing it reverses a key kidney stone risk factor. In trials potassium citrate reduced stone formation.
Potassium Citrate is Preferable to Sodium Citrate
I have a long list of sodium’s undesirable effects. It can raise blood pressure in large numbers of people, especially with age. It raises the amount of calcium lost in the urine, and that increase of calcium can raise supersaturation and promote calcium kidney stones. High sodium intake can reduce bone mineral retention. But, it may be that the sodium in sodium bicarbonate causes less of these problems than the sodium in sodium chloride – table salt. So I offer sodium bicarbonate as an alternative – with reservations.
Because sodium produces problems of its own, we tend to use potassium citrate as the preferred medication, and generations of stone patients have taken it. In several trials it has reduced new stone formation when given to patients whose urine is citrate deficient.
IS THERE A COST ISSUE?
I am not at all sure why the pricing of potassium citrate has become a topic I often hear about from patients, doctors, and just about everybody in the kidney stone world. Certainly the price must have increased, but I cannot find data on the web to prove the point. I also believe Medicare and perhaps other insurers have altered the status of this drug in their payment schedules. Perhaps some of you know more about the problem than I do and are willing to share what you know by way of a comment.
I did find on inspection of the Medicare lists of drug prices by insurance plan that some plans appear to include potassium citrate pills in their formularies at a preferred level and charge as little as $10 for what appears to be 90 pills. Others do not do this and publish higher prices, often as percentages of the retail cash price. Once again, I hope those of you with experiences in purchasing the drug will share what you know.
CAREFUL SHOPPING LOWERS PRICES
Listening to agitated, and worrisome stories about inflated prices for potassium citrate, I decided to try to be helpful. A Google search for prices of potassium citrate yielded a few promising shopping sites, and on study of the prices I found some much better than others. Note that in the following sections I present a lot of prices and arithmetic. Sometimes, when the message is very clear the results are rounded for simplicity. I give the basis for every calculation if you want absolute exact answers to the nearest penny. Likewise, because we are comparing prices, I have chosen 4 pills daily as my cost basis. The actual range can be from 2 to 6 pills or even more daily, so you will have to adjust costs to your own prescription.
SAM’S CLUB
GoodRx gives what I believe is the clearest list of prices. On their site, Sam’s Club was least expensive at $145 for 180 pills or $0.805 per pill. A typical 4 pills per day treatment option would therefore come to $290/quarter, which is still very pricey. The site gives a long list of other stores whose prices are even higher. Everyday health offers an approximate price for Cytra-K and Polycitra K of $50 – $99, but I could not be sure if this was for a month and likewise how much medication was in a dose.
CANADA
So far as I can tell, importing from Canada will not save you much money. I found Urocit K at $1.10 per tablet, which is higher than Sam’s Club. Another generic, K-Citra 10 was $0.79, which is about the same as Sam’s Club. Another less desirable canadian price was $0.52 per pill if you buy 90 pills, but it was for the 5 mEq size, 1/2 of the usual and therefore the corresponding price for 10 mEq would be $1.04/pill. Given that some costs must accrue for mailing, and there are issues with importing, I cannot see an advantage right now.
WHAT TO DO
Shop Well
Certainly web shopping is a good thing because in my modest and amateurish shopping efforts I found a tremendous range of prices. I am sure that many of you who read this post are far more skilled than I am at shopping for best prices. It is time for you to step forward and share your knowledge with all of us by posting a comment. Everyone will benefit and appreciate your contributions.
But even if you shop better than I did, retail pricing for this medication seems too high for most to afford. At even 4 pills a day, and at the best price I found ($290.00/quarter) we are over $1000.00 yearly for this one product. It seems to me that if your plan does not subsidize this medication, cost could be a serious issue.
Use Beverages
A useful publication reports the alkali content of commercial beverages. The ‘lemonade formula’ referred to on the graph is given as 1/2 cup ReaLemon© mixed with 7-1/2 cups of 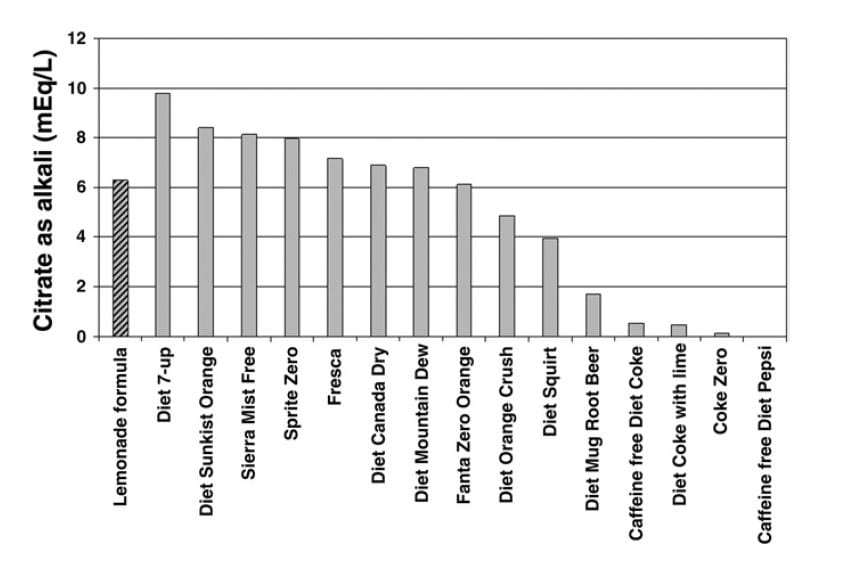 water and sweetened to taste with sugar or artificial sweetener. Diet 7-up was the winner with 10 mEq of citrate in a liter. A single Urocit K tablet contains 10 mEq of potassium citrate, as a comparison, so you would need 4 liters of the beverage daily to match 4 pills.
water and sweetened to taste with sugar or artificial sweetener. Diet 7-up was the winner with 10 mEq of citrate in a liter. A single Urocit K tablet contains 10 mEq of potassium citrate, as a comparison, so you would need 4 liters of the beverage daily to match 4 pills.
You Can Do Better
My colleague Dr. John Asplin has measured an additional group of products: Minute Maid Lemonade contains 10.3 mEq/liter of alkali, like Diet 7-up. Gatorade contains only 8.3 mEq/liter. But Crystal Light Lemonade contains 21.7 mEq of alkali, so it is the winner. Each liter substitutes for 2 potassium citrate pills, $1.60 a day, or $144 every 3 months.
We know About Classic Crystal Light
Crystal Light beverages include teas and other drinks. Our measurements refer to the classic or standard lemonade beverage. In what follows all of my remarks at bounded by that limitation. For example, I do not know if liters of the Crystal Light tea might contain excessive amounts of oxalate.
The Prices of Crystal Light
I did not research the price of Crystal Light Lemonade extensively, but Crystal Light Lemonade Pitcher Packs – 3-Pack – are $27.95 at Amazon. Each 3 pack provides 96 quarts of beverage. Each quart is about one liter (0.946 liters to be exact). The cost is therefore $27.95/96 or about $0.29 per 20 mEq (2 pills). This comes to $0.58 daily or $52 every three months. The Amazon site points out that prices might be lower at other stores. Please comment on the best prices you have found so everyone can benefit.
It is Not Just How Much Citrate is in the Beverage
You may have read, on a label or in a scientific paper, that some of the beverages I have listed contain quite a lot of citrate, yet we show them as inferior as an alkali. The reason has to do with the form of the citrate. If the drink is made up in a very acidic manner, much of the citrate is citric acid and will not produce alkali in the body when metabolized. It is only when the molecule is citrate itself, not the citric acid, that it can benefit you as an alkali. The graph and the additions by Dr. Asplin present the true alkali content.
Be Wary of Sugar
The beverages are mainly diet so they do not add to your caloric burden. If you sweeten them, or lace them with fruit juice, or add fruit juice or other flavorings to baking soda – see below, you will be adding calories to your diet and that may not be ideal.
But apart from weight gain, sugar has undesirable effects specific to kidney stone formers: It raises urine calcium losses. Even worse, as the article points out, urine flow rate falls as urine calcium increases, so supersaturation rises for two reasons.
What About Sodium Bicarbonate
It Has a Lot of Alkali for the Money
Baking Soda
According to Google, a teaspoon contains 4,500 mg of baking soda (sodium bicarbonate). Given the molecular weight of 84 mg/mEq (each molecule is one mEq of alkali) the teaspoon contains 53 mEq of sodium alkali. In principle, therefore, one can get alkali for nearly nothing by way of price. According to Dr. Asplin, who has – unbelievably – determined such matters, a teaspoon can contain up to 6,100 mg of baking soda depending on packing and whether the teaspoon is level or heaping.
To get 20 mEq of alkali from baking soda would require about 1/3 teaspoon. Given the variability of what a teaspoon holds, and the sheer problems of fractions of a teaspoon for every dose, I strongly recommend we abandon the remarkable cost savings from baking soda and use sodium bicarbonate tablets, which are very inexpensive and measure out the dose for you.
Sodium Bicarbonate Tablets
You can buy sodium bicarbonate tablets OTC and they are cheap. Concord, via Amazon, sells one hundred 650 mg tablets for $14.95 ($0.14 each). Rugby sells 1000 tablets of the same size for $25.77 ($0.026 each). Because each tablet contains only 7.7 mEq of alkali, it takes about 3 to match 2 K citrate pills (I realize 7.7 times 3 is 23.1 mEq but it approximates 20 mEq and the difference is not important). But that is only $0.075 for the three. So the price can come way down with this form of alkali.
It has a lot of Sodium, Too
But, alas, the 1/3 teaspoon, or the three 650 mg pills, deliver 20 mEq of sodium for each 20 mEq of alkali. The extra 20 mEq of sodium is 460 mg, about 20% of a full day’s sodium intake. For the 40 mEq (4 potassium citrate pills) we have used as a benchmark thus far, it is 40% of a full day’s sodium intake.
Whereas I am unconcerned to recommend beverages as replacements for potassium citrate pills, I have considerable reservation about sodium loads for reasons I have already mentioned and repeat here for emphasis. Excess sodium intake can raise blood pressure in those who are sensitive to salt. Although we have not as yet discussed urine calcium losses as a risk factor for stones, sodium loads will raise urine calcium, and are therefore not beneficial in that respect. If you are taking a diuretic to reduce urine calcium for stone prevention, sodium loads will reduce the efficacy of the treatment and promote losses of potassium. People with heart disease may develop worsening heart failure. Always ask your physician before using sodium bicarbonate as an alkali.
Even so, sodium bicarbonate is not sodium chloride – table salt. For physicians I have reviewed a few papers on the subject. If I sound ambivalent, I am. We may need a few more trials on this subject. In the mean time, all of my reservations hold sway. Use sodium bicarbonate sparingly.
How To Put It All Together
Compromise is the best policy, and I offer a general scheme which patients and physicians can use, if they wish, with their personal alterations. Be sure and check that your combinations provide the dosages your physician wants you to have.
Make a List of Equivalent Dosages
Each potassium citrate pill is 10 mEq; 2 are 20 mEq of alkali. Each liter of Crystal Light is just over 20 mEq of alkali. Each OTC 10 grain (650 mg) sodium bicarbonate tablet is 7.7 mEq of alkali so 3 make 23 mEq.
Make A Day’s Menu
Consider dividing the day’s alkali into 3 parts: Beverages; sodium bicarbonate; potassium citrate pills.
To Replace 2 Potassium Citrate Pills
If we only need 2 10 mEq potassium citrate pills (20 mEq), substitute 1 liter of Crystal Light (20 mEq). It is part of the day’s fluids, but also like a medication, so spread its use out over the day and, if possible, night.
To Replace 4 Potassium Citrate Pills
If we need 4 pills (40 mEq) consider 1 liter of Crystal Light and three sodium bicarbonate pills (20 mEq). The beverage and individual pills can be spread out through the day.
To Replace 6 Potassium Citrate Pills
If we need 6 pills (60 mEq), consider 2 liters of Crystal light (40 mEq) and three sodium bicarbonate pills (20 mEq) likewise spread out through the day. Reserve the potassium citrate pills for when you tire of the beverage or if the extra sodium is raising blood pressure or urine calcium.
Use Many Beverage Types But Keep the Dose of Alkali The Same
Crystal Light is convenient because of how much citrate it contains. But the chart shows many alternatives which can be used instead in larger volumes. Just remember to multiply so the total amount of alkali remains about the same. For example, you need 2 liters of Diet 7-Up to equal one liter of Crystal Light.
Be Inventive: Not All Days Need Be The Same
Mixing and matching is perfectly acceptable. Each day need not look like the one before so long as the correct amount of total alkali is used. The only drawback of a mix and match approach is confusion, so make lists and keep track. As a general rule, try to make the sodium component smaller than the beverage component. Keep the expensive potassium citrate pills as a convenience and source of variety. Obviously if sodium is contraindicated medically, and beverages are too tiresome as a source for all the alkali that is needed, potassium citrate pills can be used to replace sodium bicarbonate pills.
Not All Patients Need Potassium Citrate Or Any Other Alkali
This post is for those who have been told by their physicians to use alkali. Nothing I have written here should induce anyone to begin alkali unless their physician has prescribed or recommended it. Stone formation is complicated. Sometimes alkali can worsen stones, or even become a danger. Potassium can itself be dangerous if kidney function is below normal. Sodium loads are a problem for people with high blood pressure, heart disease, and other illnesses. Do not use sodium or potassium alkali or even high volumes of Crystal Light unless the physician who is treating your stones recommends you do so.
Stay Hopeful
Whatever caused the price rise, the changes in how insurers pay for this medication, or both, may be transitory. Millions of people have kidney stones in the US. Prices for 90 days of a standard treatment are so high that few can afford them without serious budgetary concerns. When so many people are affected, hopefully market or even political forces will countervail. In the meantime, between a few potassium citrate pills, a few liters of Crystal Light, and maybe some sodium bicarbonate, physicians can piece together an adequate regime of alkali for those patients who need it. Not every stone former does need alkali, of course.
ANOTHER AND FINAL DISCLAIMER
I have brought Crystal Light to your attention as an inexpensive substitute for some of the medicinal alkali your physicians may have prescribed. As in my initial ‘Loud Disclaimer’ I say here that I receive no financial or other benefits of any kind from the makers of this beverage, have not, in fact, ever tasted it, and do not currently plan to do so. My evidence for the value of Crystal Light comes from the work of Dr. John Asplin, and comparisons to the published work of Dr. Eisner and his colleagues.



Potassium citrate was prescribed for me. I was very allergic and had to stop. Crystal light also caused hives. My citrate level was less than 100. Several months later I had PCNL. My kidney doesn’t function due to the stone that may have been there a decade. The surgery was due to my multiple UTI’s . Retested 8 weeks after surgery. 24 hour urine now shows normal citrate level for someone with one functioning kidney. Around 400. I made no changes other than following kidney stone diet. No increase in citrus. How could this happen? Results are now “ no metabolic disorders” . Thank you.
Hi Dana, I gather you have lost a kidney from obstruction and had many infections ascribed to stones in that kidney. Your urine citrate was very low and then after removal of stones from the damaged kidney citrate rose to near normal. There are two explanations, perhaps more. Firstly, bacteria in your urine can metabolize citrate so it is low when measured. A more interesting possibility is that you were ill when infected, and possibly has low levels of blood borne infection. That will cause your cells to produce excessive acids, and kidneys will respond by reabsorbing citrate so it does not appear in the urine. Of course lacking details I cannot tell one from the other, and perhaps there were even other factors at work. But these are good likelihoods. Regards, Fred Coe
I was prescribed potassium citrate pills 1620mg (15meq), but they are rather large and sometimes cause me to gag. I have no problem swallowing other pill medication. Do you know if they come in generic form in smaller sizes?
Thank you
Hi Marissa, Indeed the usual size is 1080 mg, and one can just use more of them to get the same doses. Check with your pharmacy or physician. Regards, Fred Coe
I recently had one stone lasered and they found two more in my kidneys that were not treated. I am a 72 year old mail and my doctor has prescribed two 10 mEq potassium citrate pills per day. I was able to get 180 pills at Costco for about $80. That was after Walgreens wanted $210 for the same thing. Checking Costco might be helpful to others. My problem is not the cost of the pills, but that I can’t seem to swallow them. I have no problem with other pills, but these things get stuck in my throat I have become very afraid of taking them for fear of choking. Are there any brands with a smooth coating or is it possible for me to coat them with something that will make them not stick to my mouth, tongue and throat?
Hi Bob, Many people have this problem. I know of no alternative brands for the plain K citrate pill, and I am not enthusiastic about coating them. There are liquid alkali products, and the best source is your pharmacist. She/he is permitted to recommend alternative formulations to provide a specific dose of medication. Regards, Fred Coe
Like many others, I cannot swallow potassium citrate pills and I am not a fan of aspartame. Is Litholyte an acceptable substitute? it is 10mEq citrate and bicarbonate. it has 200mg of potassium citrate, 30mg of magnesium citrate, 60mg of sodium bicarbonate and erythritol.
Hi Mary, It has abundant sodium and bicarbonate is absorbed and acts rapidly as opposed to citrate that is metabolized to bicarbonate. But it is better than nothing assuming you need extra citrate – be sure. Regards, Fred Coe
With Covid 19 my surgery date to remove an 11 mm kidney stone was canceled. I can feel it and while it’s not causing horrid pain I’m seriously concerned. My urologist suggested adding more citrate to my regimen to prevent further growth but I had to cut daily fresh citrus intake since my stomach just can’t handle it. Which product route would you recommend? Thank you.
Hi Karen, Citrate has been used in a few trials to slow growth of stones after surgery with positive results. Your physician can prescribe medicinal potassium citrate if she/he believes it will help. Regards, Fred Coe
I notice that Amazon sells large bags of Potassium Citrate with no additives. I have not worked out the cost per day equivalent to 10 MEq TID. Do you have any information about taking the medication that way?
Hi Jon, If you read back over the comments on this post you will find – probably – over 100 about people doing it. Most buy a home scale accurate to 0.1 gm or less and weigh out 1 gm doses – 10 mEq tablets contain 1080 mg (1.080 gm) of the crystal. It is fine if your physician knows and approves, you have the right scale and know how to measure accurately, and do not mind the extra work. But be sure your physician knows and approves, potassium can injure some people who have reduced tolerance to it. Regards, Fred Coe
Hello , Do you use in your practice the over the counter postassium citrate 99mg for your patients. Do you know how much citrate these contain? Thanks in advance Missy McClenahan PAC
Hi Missy, a 10 mEq tablet (single dose) of potassium citrate is 1080 mg, so 99 mg is about 9.2% of that. So you would need about 9 of the small ones for one full dose, and most people need 2 -3 doses /d, or 18 – 27 of the small pills. It can be done but not practically. Regards, Fred Coe
I also could not swallow the Potassium Citrate 10 mEq pills either so my Doctor prescribed me the 5 mEq size which I swallow easily with a spoonful of jello and can take 2 each time to get the 10 dose. They are about the size of a thick Advil.
Hello Dr. Coe,
First, thank you very much for this information.
How many times can we dose per day? Is there an upper limite. For instance, I prever the 99mg pills and realize I need 10 to make a single dose. I don’t mind taking this multiple times a day and am fortunate to be able to do so. Would I split the doses up between 6 hours? Say, take 99mgx10 pills at 8am, 2pm, 8pm? Or would it be better to dose it 6 times a day and split the doses in half?
Best,
Srdjan
Hi Srdjan, 10 mEq of potassium citrate is a standard unit dose and common starting doses are 20 mEq twice a day, but I am unclear as to what you are treating. Do you have calcium oxalate stones, calcium phosphate stones, uric acid stones? Does your urine have a low level of citrate, or too low a pH? One does not use potassium citrate except for a purpose and I am not able to determine that purpose from what you have written. Regards, Fred Coe
Dear Dr. Coe:
I read your comments & speculation as to why potassium citrate tablets appear now to be so expensive. I have some relevant comments about the cost: I have been taking two tablets/day, of dosage 10meq (1080mg by my calculation) each for three years now on prescription from my urologist. Prior to the three years, I had a history of 8 stones in 10 years, all calcium oxalate, including one in each kidney at the same time that were removed by surgery. While on employers medical insurance, I paid about $3-5 for a 90-day prescription of 180 tablets. These tablets are composed of 10meq of powdered potassium citrate encased in an extended-release carnuba wax, according to the manufacturer Zydus Labs. I note here that the release rate is not specified nor guaranteed and that another manufacturer of pharmacy/physician controlled potassium citrate tablets also does not publish a specified or guaranteed range of extended release rate.
Since recently retiring and now on a Part D drug plan, I was recently quoted $300+ for a 90 day prescription (180 tablets of 10meq) with a $300 annual deductible, because the medicine is classified as a Tier 4 medication. I found the tier classification to be highly suspicious, because potassium citrate is a simple inorganic compound with a chemical structure of the same general complexity as aspirin and with an equally simple method of manufacture. i have discovered that powdered potassium citrate is widely available online (Amazon, etc) and in Health Food stores, but this non-prescription format is powdered in gel capsule format, meaning that it is quick release. The maximum dosage per gel capsule is specified as 270mg (2.5meq by my calculation), so that I would need to take 8 capsules per day. I have also discovered that potassium citrate is available in bags of 1kg each, all from FDA certified labs and none by prescription. Well I just purchased a year’s worth of gel capsules for $61 on Amazon and am looking forward to taking two capsules 4 times per day. I think my chemistry is correct but would appreciate your comments. Do you know if CMS dictates the Tier classification of the formulary or leaves that to each Part D vendor?
Best regards, Charles Breverman, charlesbreverman@comcast.net April 14, 2020
Addendum: Note that some of the non-prescription capsule distributors specify the potassium citrate capsules as having a dosage of 99mg or 100mg. This is the mass of only the potassium content. The total weight of potassium citrate should be 270mg which again I calculate to be equivalent to 10meq which is the published dose for the two brands of pharmacy/physician controlled tablets encased in caruba wax.
Hi Charles, 10 mEq of potassium citrate crystals is 1080 mg. The crystals have 3 potassium atoms per citrate molecule because citrate has three negative valence sites. So it takes 3 x 39 + 192 = 117 + 192 = 309 mg for 3 to make a single 3 potassium/one citric acid complex that has 3 mEq. To get 10 mEq takes 10/9 x 309 x3 = 1030. The 1080 comes from additional water molecules in the potassium citrate crystal. As for using FDA approved potassium citrate I see no problem except that you need to weigh it out. Your physician must be a part of this, for safety. Regards, Fred Coe
Dear Dr Coe:
I recently wrote to you questioning how important is the difference between the release (dissolution) rate of taking two 10 meq (1080mg) hard tablets (encased in carnuba wax advertised as extended release) per day of pharmacy/physician-controlled potassium citrate versus taking two gel capsules 4X per day of 2.5meq (270mg) OTC potassium citrate or even taking bulk anhydrous powdered potassium citrate at the same quantity and frequency. I note only that two of the manufacturers of the pharmacy/physician controlled hard tablets do not specify what the extended release rate is and therefore are not guaranteeing a release rate. Your comments still much appreciated.
I read the latest comments from a Missy McLenahan on 3/20/2020 wherein she assumes incorrectly that a bottle of 99mg capsules offers a per-capsule dosage of only 1/10th of the dosage of 1080mg or 10meq tablets. Therefore she incorrectly concludes she needs to take 10 capsules to get the 1080mg of citrate. In fact each gel capsule contains 270mg of citrate or 99mg of potassium which offers 2.5meq per capsule. Please correct her understanding else she is looking at an overdose. Thank you.
I have just written a story of this whole potassium citrate pricing lunacy to Scott Gottlieb, former FDA Director, who is a friend of a friend. He has a public email address at the American Enterprise Institute. Maybe he can explain.
Hi, It is not so much a lunacy and a form of price gouging. The intermediary companies that market drugs are much responsible as they get a fraction of the final price. Altogether the K citrate story is a small example of US wide pricing problem. The other side of the problem is generic drugs whose cheapness depends on manufacturing in India where quality control has been shown non existant or poor, deceptions the rule, and drug component often from China and adulterated. If were young today I would have aimed my career at the interface of medicine, business and government in hopes of improving matters. Too late for that! Fred
Dr Coe,
Thank you for your work on this subject. After suffering kidney stones annually for thirty years, in 2016 I read your paper published in 2010 and worked with my urologist to implement your findings with great results.
I am taking 2 – 15meq tablets of Potassium Citrate daily and now am being advised to increase the dosage to 3 tablets daily as a result of urinalysis. I am drinking a gallon a day of water and would prefer , for cost reasons, to take the additional dose by just dissolving some of FDA lab supplied powdered potassium citrate in the water I drink daily.
Do you know the solubility of PotCit in water? Also, using earlier comments in this stream, I would need 1620 mg daily to get the additional 15meq tablet equivalent. Is my math correct?
Of course, I would have to get the scale and check with my physician before doing this.
Regards and be safe,
Ken Krummrich
Hi Ken, Many people are measuring out K citrate, and it is not unreasonable. The amount is correct. Likewise that you need a proper scale and your physician as an allay. Fred
Hello Dr. Coe,
Thank you for the explanation.
In your list of equivalent dosages, you wrote “Each potassium citrate pill is 10 mEq; 2 are 20 mEq of alkali. Each liter of Crystal Light is just over 20 mEq of alkali. Each OTC 10 grain (650 mg) sodium bicarbonate tablet is 7.7 mEq of alkali so 3 make 23 mEq.”
Is the alkali load provided by citrate similar to that of bicarbonate? I thought that one citrate molecule is converted to 3 bicarbonate molecules, and therefore, the actual alkali load provided by 10 meq of citrate is 30 meq of bicarbonate, which equals to 4 tabs, or 3 grams of sodium bicarbonate. can you please solve the confusion?
Hi Dian, The 10 mEq means ten milliequivalent, so the moles of citrate have been multiplied by 3, one for each valence site. Bicarbonate has a valence of 1, so one mole of bicarbonate is one equivalent of alkali. Fred
Our state employee insurance plan recently changed potassium citrate to the status of non-preferred generic, more than doubling the co-pay price we have to pay. So now I’m paying more for that than I do for a name brand drug like Brilinta, which I also take.
Hi Dr. Coe, hope you are doing well. I will be turning 65 soon and looking into drug plans. I have been taking K-Citrate for years now. 10meq 4 Tabs 3 times a day. I read your article and looking for alternatives. Anything new with Cysteine stone meds. Thank you
Hi Cathy, I hope you are doing well. Many people are buying food grade K citrate and weighing out 1 gram doses. If your present physicians agree to supervise, this is not a bad idea. I am doing well, and hope the same for you. Warmest regards, Fred
My wife takes 4 Potassium Citrate tablets daily for kidney stones (1620MG [15MEQ]) A 90 day supply is $255. For Potassium Citrate, I think this is completely unreasonable. We are on Medicare and actually have a Medicare supplement plan and this is what we are charged. When one considers the cost of hospital stays or ER visits for kidney stones, it would make sense for all parties to get the price of this product down. It is not like a lot of research went into the development of this formula. Prescription drugs are without a doubt a huge cost for us elderly and one of the problems is a drug like this that doesn’t cost that much to manufacture it is absurd.
Hi Bill, All true and we all agree. Many have gone to food grade potassium citrate and a home scale to weigh out the dose. The price falls to near nothing. But if she does this her physician needs to know and to agree. Regards, Fred Coe
I am 56 years old and have been a prolific former of calcium oxalate stones for my entire adult life. (The first radiologist to see my kidneys suggested that the stone pattern was indicative of medullary sponge kidney, although that diagnosis has never been definitively made.) I have for most of my life drunk water almost exclusively, with very little juice, soda, alcohol, etc. Over the past few years, I have been trying to consistently “spike” my water with ReaLemon (or equivalent) reconstituted lemon juice, to the tune of about 2 to 3 ounces a day. (I.e., I try to get through a quart bottle in less than 2 weeks.) It definitely does seem to be helping slow or stop the progression of the stones. I personally find no need to sweeten it in any way and, in fact, prefer this lemon water to plain water. (This is fortunate because I do not tolerate the aftertaste of any artificial sweeteners, so if I did need it to be sweetened, I don’t think I could find any alternative to sugar.)
Hi Joel, I sense from your note that you have not thus far been fully evaluated for the cause of your stones, and if I am right here is my best for that purpose. Water alone is next to useless as a stone prevention, lemon total nonsense. Take a look and see if you have been evaluated and if so what was found and how to exploit findings to create really powerful prevention to stop the stones. Regards, Fred Coe
Hi Dr. Coe, what do you think about Potassium Bicarbonate (available OTC) instead of Potassium Citrate?
Hi Josh, Shorter acting, so one wants to take it 4 times a day. The dose form is usually 3 mEq of potassium, whereas a standard pill dose is 10 mEq, so you need a lot of them. Check that point. Fred
Hello Dr. Coe, I have recently been diagnosed with Stage 3B CKD. I am 67 years old. I have no symptoms. I have had an ultrasound that notes a stone in one kidney and a cyst in the other. Based on a 24 hour urine that suggested I be prescribed Potassium Citrate 10MEQ, 2 x day. I am confused that I see that sodium, potassium, phosphorus and protein should be limited yet I am being prescribed potassium citrate. I can understand the K citrate for stones but is it contraindicated with my Stage 3 CKD? I would appreciate your thoughts. Thank you. Steve Silverman
Hi Steven, I gather you have a symptomless stone in one kidney, and reduced eGFR. Perhaps the stone is presumed uric acid – your physicians could tell from reduced radiographic density. Perhaps your 24 hour urine pH is low, in the range below 5.5, common enough with age and reduced renal function. Of course I do not know if these perhaps are true, but your physicians know and I would ask them. If so – uric acid, low pH etc, K citrate is not a bad idea, but they have to be careful that your serum potassium remains normal despite the extra intake. As for phosphate restriction, it would be independent of the citrate. Regards, Fred Coe
Dr. Coe,
Thank you very much for this post, it improves my health because I am prone to kidney stones so i know what to do now, I was told by Dr to drink a lot of water avoid eating spinach, strawberries (and I can’t remember what else), otherwise I’ll be back in hospital in 10 years time.
Hi Nayla, I am not sure if you are getting the best treatment. Take a look here and be sure you know why stones form and if you are doing all you need for prevention. Regards, Fred Coe
I just wanted to write a quick comment about the cost of potassium citrate. In my city without insurance it will cost $500 from my local walgreens. With very good insurance it will still cost $100 Co-pay.
I found out about the way the pharmaceutical industry works and to ask for the “cash price” which is something they aren’t allowed to tell you unless you specifically ask.
I also discovered goodrx and will be using this to save tons of money. My prescription went from $500 down to $138 and its as little as $57 at other local pharmacies
Thank you, Nick. I never heard about the cash price. Fred
I have been paying this year $172/90 day supply, and I take 3 x 10 mEq a day. We are almost in September, and my pharmacy called to tell me my refill is coming to $333 for the same amount! I called Caremark (my Rx plan through work) and they validated that the “negotiated rate” went up. I have not met my deductible, so I’m stuck with $333. I will try the “cash price” trick mentioned on another comment, but this is insane.
I am having surgery for my second uric acid stone in 8 months. The first was 7 mm and this one is 4 mm but I cannot seem to pass it. I am really trying to stop this stone formation cycle but read about a lot of negative side effects of potassium citrate. It is hard for me to wrap my head around the idea that drinking a liter of Crystal Light is good for me. It has sodium citrate as well as artificial sweetener and coloring. You are one of the few doctors that talk about diet and prevention for uric acid stones so I value your opinion. Also, at 178 pounds the formula says I need to limit protein to less than 3 ounces a day. That will be tough one for me but I am motivated.
Hi Martin, Uric acid stones simply cannot form when urine pH is above 6, and potassium citrate is a safe way to achieve that pH. Here is a good review of uric acid stones. Here is a more detailed one. Since potassium and citrate are already in blood and urine, and since massively higher diet potassium than our usual is healthy, and since citrate is simply a metabolic intermediate, I cannot imagine a serious problem from the material. Crystal light is simply a diet beverage that had a reasonable amount of citrate – right pH and ample citrate. Diet has no role for uric acid stones, and reducing protein is unhealthy and basically foolish – it will not work and is holdover lore from a more ignorant era of medicine. You have curable stones using a safe remedy. If you do not want potassium citrate, sodium bicarbonate will also work at an equivalent dose, but if you have high blood pressure the extra sodium could be an issue – even then one can get around it. Regards, Fred Coe
I have been advised to take potassium citrate as part of an overall prevention protocol for calcium oxalate stones and can purchase it in immediate-release form at almost half the cost of timed-release (ie. $0.15 per 10 mEq vs $0.27 per 10 mEq). Does it matter from an efficacy standpoint what form is ingested? Can immediate-release have any detrimental health implications in the short or long-term due to absorption of the potassium citrate too rapidly (versus timed-release)? Thank you.
Hi Michael, NO data but if you use immediate release you can simply spread the dosage more – perhaps 4 times a day. K Citrate is not known to cause diseases. Regards, Fred Coe
I just just started potassium citrate twice a day. My history: bilateral staghorn calculi diagnosed in May 2020, the larger of which was 9 cm. I had pcnl on each and shockwave on the inoperable section of the larger one. Fluoscopy was clear when all the procedures were finished. I’m already growing new stones only a month after the the nephrostomy tube was removed in August. I have stage 2 ckd, high blood pressure, and possibly diabetes. Taking amlodipine. Urologist prescribed hydrochlorothiazide and potassium citrate when the CT scan came back last Thursday. I’m working on my diet, but I’m overwhelmed. The diet for high bp, diabetes, and ckd are somewhat similar, but the diet for kidney stones is in such contradiction to those.
Hi Monica, all depends on the stone type. Was it struvite, uric acid, calcium based? I would guess struvite, but you actually know. Each has its own special treatments. Regards, Fred Coe
Thank for the exhaustive research and sharing. The cost for 3 mo tidwas $442. Fortunately I have insurance. This cost is ridiculously high. Kind of like insulin that companies know you need.
Jane, it is a scandal, and no one can fix it. Fred
So are we to understand that Crystal Light has more citrate and real lemon juice?
Hi Brian, The pH is higher so a higher percent of total citrate species are citrate anion vs. citric acid. Fred
I have found potassium citrate powder for $13 for 13 oz. but i’m having trouble calculating a 10 mEq dose into teaspoons of this powder. I plan to dissolve it into water and take it 3 times per day, per my doctor’s prescription.How many mEqs are in a teaspoon?
Hi Jeff, One requires 1080 mg/10 mEq. You need to use a scale that can measure out this amount, a teaspoon is not ideal. Your physician has to agree that you can execute his/her Rx using a scale and the commercial powder, and that the powder is approved for human consumption. Regards, Fred Coe
Here is 450 doses for $10 – that would be 2 cents a dose, right? – https://www.pipingrock.com/potassium/potassium-citrate-powder-16-oz-454-g-bottle-15411?prd=D0000J&prisp=1
About 10 years ago the cost of my potassium citrate pills (Urocit-K) were getting to be upwards of $75 a month, which was just too much. I did some research and found Effer-K. It’s a dissolvable tablet that at the time I’m pretty sure was outright referred to as potassium citrate (it’s website largely still calls it that) but now has the more-wordy description of “Potassium bicarbonate / citric acid effervescent tablets” that once dissolved “provides (dosage) of potassium in solution as potassium citrate”.
A month’s supply of Effer-K cost around $12 (it’s around $16 now) compared to the $75 I was paying. I switched and haven’t looked back and after 10 years haven’t noticed any change as it relates to my kidney stones.
Hi Rex, I believe Effer-K is potassium bicarbonate, not citrate. It will have a shorter duration of action, but multiple doses per day is common. Not a bad alternative. Thanks, Fred
What do you think of the new product Moonstone? One packet mixed with 16oz glass of water contains 30 mEq 30 of alkali citrate. Ingredients on the package are magnesium, sodium and potassium citrate with sodium being 95 mg per packet. Seems to be in partnership with the National Kidney Foundation and a patented product.
Hi Michelle, I could not find the details of the product on their website, so I do not know what is in it. I do not know what the partnership means, either, but apparently the NKF does not endorse it. I imagine it is safe. If sodium is 95 mg that makes about 4 mEq whereas a packet is said to be 30 mEq of alkali. Confusing indeed. Regards, Fred Coe
Dr. Coe, there are many powders, pills, drinks, etc. that advertise their product as potassium citrate for very low prices (e.g. $5 for 180 pills). However, when I review the ingredients the labels state “Potassium (from Potassium Citrate)” or “Potassium (as Potassium Citrate).” Are these products misleading consumers by stating they are potassium citrate when they are actually just potassium? Or is “Potassium (from Potassium Citrate)” a legitimate active ingredient we are seeking? Thanks for your help!
Hi Jeffrey, the phrase means – or better mean – potassium citrate; there is no potassium without an anion – chloride, phosphate, citrate, bicarbonate, etc. Fred
I see there are lots o dietary supplement pills on the market, both the potassium magnesium citrate and potassium citrate. For potassium magnesium citrate the dose in mg is 70 for potassium and 140 for magnesium. Would 5 pills be approximately 10 mEq?
Hi Carl, These supplements vary so I can only help if you specify which one(s). Best, Fred
If you are worried about the sodium load from baking soda, is there a reason why potassium bicarbonate might not provide a similar benefit. You can get gelatin or vege caps that could probably easily hold a daily-supplement amount in a few caps, and you can buy the potassium bicarbonate online by the pound — it’s sold as an additive for wine makers to adjust the ph of wine. Also with so many foods containing added sodium and so few containing potassium, I substitute ‘NoSalt’ (Potassium Chloride) where-ever a recipe or mix calls for adding salt. Even though I am overweight by more than a bit, my BP is still in the healthy range as well as my blood sugar.
I have no idea what the therapeutic dose for Stevia is for lowering BP, but I use it for many-most of my sweetening needs,. Just so happens one of my favorite drink mixes includes Crystal Light raspberry-lemonade. I add a bit of that to a flavored soda water + add a few ounces (varies) of some fruit juice (like raspberry-lemonade). I mix it by the “serving” (~750ml), and vary the proportions by taste.
Hi Astara W, Unless your physicians are concerned about potassium intake, there is no reason you cannot use it as substitute for sodium. As a general fact, our ideal diet is much lower in sodium (1500 mg, 65 mEq) and much higher in potassium (4,000 mg, 100 mEq) than the present US diet habits. As for Stevia, I do not consider it a treatment so much as a flavoring agent. I found this review of several trials using what appears to be the plant. It is a miserable journal and I cannot get the full text without paying for it, so I am not sure about the quality. This excellent journal reports an actual trial showing a measurable increase in BP with Stevia plant extract and shows it is from corticosteroid effects. Synthetic stevia may not work the same as the plant material. Regards, Fred Coe
thank you I just got prescribed this medicine. when I went to pick it up today they wanted $235 for 90 day supply. WIth good RX it was $81 for 90 day supply. This is expensive so I am thinking I might try to stretch it further with substituting some days with the Crystal Light.
Hi Laura, I believe it is you I answered already. Be sure your testing means this is the right treatment for you. Here is yet another article to help. Regards, Fred Coe
Dr Coe- is crystal lite a safer substitute then potassium citrate for those of us who have CKD 3A?
Hi Frank, If you need potassium citrate for stone prevention – usually uric acid stones in people with mild renal impairment – routine potassium citrate is safe. But I do not know your details, so this is mere commentary. Your physician is responsible for your safety. Regards, Fred Coe
Potassium citrate is listed in diet lipton lemon iced tea mix,but I’m wondering how much. Is there any information on how much potassium citrate is in a prepared liter of this tea mixbor something similar?
Hi Bella, Potassium citrate is common in food processing. The amounts will be medically unimportant if you actually need that material for stone prevention. Fred
Dr.Fredric Coe;
After my 2nd kidney stone and my 3rd 24hr urine test analysis, ( previously was told to take potassium citrate pills but the cost was prohibateive) -my urologist recomended LithoLyte ( 1 packet in the morning and 1 packet in the evening ie 2 /day). With the auto ship program from the company the cost before tax would be $43.99/ month. This was a 50%+ savings over the Rx. After seeing your article, i could use Crystal Light (2 packets a day same as my doc’s recomemendation but the cost would be $14.88/month if this were the only usuage. i am thinking that a amajority of Crystal light with an occassional 1/3 teaspoon of sodium bicarbonate-trying to be “inanovative” as the article suggests. My 24 hr test show not enough urine output I thought it was 2liters but for some reason the lab recoreded the output as 1.5 liter. i pee all the time !! At any rate my 3 tests show a decrease from 2.0L to 1.66L and now 1.5L currently. Any suggestions about how i can get more urine output ( or possible problems causing output?). I had be switched from HTZ to Amlodipine 10mg for hypertension by my primary MD. I had notice a swelling of the ankles which the primary MD says is typical . I also take Atorvatin Calcium 40mg /day. Thanks for your insightful and thorough research and presenting this factor in an understanding way for the general population.
Hi Paul, The urine volume is the excess of water intake over losses from sweating and respiration. This means you do not drink enough. Frequent urination in man often means prostate enlargement with a low capacity bladder. Stopping the CTZ was related to your BP, but was it also lowering your urine calcium as part of stone prevention? When you stop a diuretic there is transient sodium and water conservation – I hope you did not collect your 24 hour urine during that transient phase of about 2 weeks! Why are you taking citrate, is your urine citrate low, or is your urine too acid and you form uric acid stones? IN other words, what are the 24 hour urine abnormalities thought to cause your stones? Does the citrate correct them? As for your BP, if CTZ was not lowering it enough, it is usual to add a second drug to the first, to get synergy. Lacking direct knowledge of your situation, please consider these outsider remarks, as they may well be off the mark. Regards, Fred Coe
Hi Dr. Coe,
I was wondering why there is no mention of simply using lemon juice as a source of citrate. According to the article linked below, “Approximately 85cc of lemon juice contains 60 mEq (4.2 gm) citrate.” Is there any reason to not add ~1 lemon worth of juice to water over the course of a day as an alternative to potassium citrate?
Cheers!
https://journals.lww.com/jbisrir/fulltext/2011/09481/the_effectiveness_of_lemon_solution_versus.8.aspx#:~:text=Approximately%2085cc%20of%20lemon%20juice,equal%20as%20comparator%2Dpotassium%20citrate
Hi Nadim, Many have used it. If citrate is desired, the amount in a lemon will vary with the variety and ripeness. Crystal Light lemonade is cheap and one liter provides 20 mEq of potassium citrate. Are you sure you need potassium citrate? Do you have calcium stones and low urine citrate? Be sure you have been fully evaluated and need this remedy. Regards, Fred Coe
Hi.. I just found out that an independent nearby pharmacy here in Toronto can order bottles of 100 potassium citrate pills for $5 each. They are from a company called Atoma Assurance who sell various supplements and OTC pharmaceuticals. The label/info on the bottle only says each pill is 99mg but my pharmacist did a bit of research and found they are 2.53Meq each. So doing the math 4 pills or 10.1Meq works out to be 20 cents. This is their parent company’s web site:
https://www.groupeproxim.ca/en/products-and-services/atoma-private-label/a-closer-look-at-our-products
Hi Harvey, he and you are right, others have found the same. If you need this drug, 4 pills are about 1 normal pill, so a common starting dose would be 4 pills 2 times a day. But are you sure it is the right drug? Is your urine citrate low?? Is is because urine pH is low?? It will do nothing unless it is bringing something abnormal into a more normal range. Regards, Fred Coe
Yes, I’ve discussed this a number of times with my urologist. I’ve had/passed 15 kidney stones over the last 25 years. We’ve tested a number of them and they were all 100% uric acid stones. They are usually between 3mm and 6mm. Since I started taking potassium citrate (20mEq/day) 4 years ago (and regularly test the pH of my urine) I have not passed any stones.
Thanks for your help. It’s greatly appreciated.
Oops. Pls feel free to edit Meq to mEq in my previous comment. Tnks 🙂
Thank you for providing such detailed scientific information for everyone. In this article, you mentioned that potassium citrate may be harmful for some types of stone formers. I was a little confused as I understood from your article entitled CITRATE TO PREVENT CALCIUM AND URIC ACID STONES that potassium citrate is beneficial for calcium oxalate stones, uric acid stones, and possibly beneficial for calcium phosphate stones. Could you kindly clarify in which circumstances potassium citrate would be harmful? Thank you so much.
Hi Nancy, Careful reader – I should write up to your thoughtful level. Calcium phosphate crystals form when urine is more alkaline – what happens when you take K citrate – but the drug can raise urine citrate that lowers calcium crystal formation. SO there is a kind of tension between the benefits of the citrate and the risks from more alkaline urine raising calcium phosphate crystallization. No trial has concerned calcium phosphate stone formers as a specific group – they are just a minority of ‘calcium’ stone formers studied. So, no one knows if k citrate is good, bad, or indifferent for calcium phosphate stones. I have asked my friends to do a trial, but no one thinks it worthwhile. Thanks for reading to well. Fred
Hi Dr. Coe.
Thank you for all your thorough info. I was just prescribed Potassium Citrate due to my Calcium Oxolate stones. The Rx for 180 pills is $120 at Walmart. I stumbled across your article and had a question about the benefits of Potassium Citrate for Calcium Oxolate stones or is it just for uric acid stones? It was my family physician that prescribed it and not the Urologist. I’m just wondering if I should check with the Urologist before starting the Rx, especially if it won’t help my kind of stones? Thank you in advance for your help!
Hi Laura, There are trials of potassium citrate for ‘calcium’ stones, no doubt mostly calcium oxalate stones. So the drug is not without evidence. Regards, Fred Coe
Dr. Coe,
I’ve been a member of Jill’s group for almost 4 years. In that amount of time I’ve followed the diet, added a few supplements suggested by the nephrologist (mg, K2, D3, B complex) and I’ve also added 1 liter of Crystal Light to my daily water to substitute the 10mEq of potassium citrate the nephrologist also recommended. I’ve remained stone-free all this time. My question is, my 24 hour urines prior to starting the added citrate showed my citrate levels to be adequate and in the 600s, but the nephrologist still prescribed potassium citrate as an additional strategy to lower my urine calcium. While my urine calcium’s have lowered significantly from mid 400s to 230ish, my citrate levels have risen into the 900s. I’m trying to figure out if there’s a sweet spot so I don’t overdo a good thing. Would it be smart to cut back on the Crystal Light perhaps to 1/2L? Or is my 4 year track record a good indicator I’m where I need to be? I also worry about the aspartame. Should I be?
Thank you so much for your consideration.
Hi Melissa, Aspartame does not scare me – most sugar substitute scares have arisen from Big Sugar paying scientists to find a problem, any problem, with them. The extra citrate does not scare me, and using K citrate to lower urine calcium a bit is not at all irrational. I presume your stones were not calcium phosphate as urine pH will be increased. No stones for 4 years, incidentally, sounds good, so perhaps things are about the best they need to be for you. Regards, Fred Coe
Dr. Coe,
Your input is reassuring. Thank you so very much.
Melissa Howard
Have heard or suggested Ortholyte? It looks to be a combination of pot citrate, mag citrate and sodium bicarbonate.
John, Even at page 10 on Google I cannot find ortholyte apart from shoes. Can you post a link to it??? Fred
Hi Dr Coe,
How effective is magnesium citrate compared to potassium citrate at increasing urinary citrate and Ph? Thanks
Hi Ric, Very limited because it affects bowel function. Fred
Hi Dr. Coe,
How much dosage of potassium citrate is needed to raise urine pH from 5 to 7? Thank you.
Hi Haidar, about 30 to 40 mEq daily should do it, but use 24 hour urines to be sure. Fred
Thanks Dr. Coe.
And after starting to take 30-40 mEq/daily, how long before I can expect to see the effects or changes on my urine pH, will that take few days or within 24 hrs?
Hi Haider, IN the first several days. Fred
Hi Dr. Coe,
I thought about a fun or laughable idea. As far as increasing the intake of citric acid from lemons and increasing the citrate level in the urine are concerned, what about putting crystalized lemon (e.g. the “True Lemon” brand on Amazon) in empty vegan capsules and swallowing the filled capsules with water? This would resolve the issue of Crystal Light drinks potentially causing problems to teeth as well. Also, aspartame (used in Crystal Light) appears to be controversial even though it is approved by FDA.
Hi Dynas, I am not so sure. First, is there a reason you need or want potassium alkali? Is your 24 hour urine citrate low, or your urine pH low? Are your stones calcium oxalate and not calcium phosphate? Is citrate or pH the key abnormality? Here is a reasonable introduction to stone prevention, perhaps it might be of value. Regards, Fred Coe
Much appreciated Dr. Coe.
Furthermore subsequent to beginning to take 30-40 mEq/day by day, how some time before I can hope to see the impacts or changes on my pee pH, will that require not many days or inside 24 hrs?
Hi Stefan, Effects would be visible on testing by several days. Fred
I recently had surgery for kidney stones. And have been on K+citrate for sometime. I have a hard time keeping my k+ up and to use crystal light as a substitute is new to me. Your article was very informative.
Hi Velma, But be sure it is the right drug for you. Take a look at how to think about your stone problem and decide. Regards, Fred Coe
Hi Dr. Coe,
I was wondering whether taking potassium citrate would generally make chronic constipation better or worse. Or would it have no effect on the existing constipation?
Many thanks,
April
Hi April, Usually neither, although abdominal discomfort is not uncommon. It is so variable among people! Are you sure it is the right thing for you? Take a look and see where you fit in. Regards, Fred Coe
I was recently put on 2 potassium citrate tablets daily—just before going on Medicare Part D. I selected a plan that showed it was covered. Imagine my horror when the approved pharmacy said it was $138 for a month on my Part D insurance, but only $35 for a month on GoodRx.
And to think I’m paying $22 monthly for the Part D plan that doesn’t cover this.
Still too much to pay monthly on Medicare in my opinion!
Hi KB, Way too expensive. Thence be sure why you take it. The usual reason in later age is uric acid stones, and if so you really must have the alkali. Frankly $35/ month is about what you would pay for the OTC things like Moon stone. The dose seems low. If you have uric acid stones it may be too low. Since I do not know why you are using this agent there is not much more I can offer. Regards, Fred Coe
I have had 4 kidney stones—diagnosed as calcium oxalate (2014-2021). It was determined through a 24-hour urine collection that I was not drinking enough water.
My last 24-hour urine collection showed enough urine output but very acidic urine. That is why I was prescribed the potassium citrate.
The 1-month Followup after taking potassium citrate showed a more normal acidity, so I will continue to take these expensive pills.
Hi KB, wise choice. Fred
I was just responding to the high cost of potassium citrate. I have had a 24-hour evaluation by Jill Harris. I have a history of kidney stones. First 24-hour test showed too little water. Second 24-hour test showed volume was fine, but acidic urine output, thus the potassium citrate has been added to my pill regimen.
Hi KB, If your stones were made of uric acid, this supplement is necessary life long. If you made calcium oxalate stones and if your urine citrate was low on 24 hour urine testing it is a good treatment but the urine outcome is not pH but citrate. If you make calcium phosphate stones it may not be ideal, but Jill would have known that. Be sure you know the kind of stone and your 24 hour urine results. Regards, Fred Coe
I purchase IKg pure Potassium Citrate, brand microingredients for $19, through Amazon. I take about 3gm 4 times a day, dissolved in about 2oz warm water with food. This is approximately what the Mayo clinic recommended online. I have been doing this for 9mos. My urine normally runs from about 5.5 to 6.5 pH. With this supplementation it runs about 7.5 pH, 24 hour average. I have no other health issues and do not take any prescriptions. Although this seems to have reduced my stones, it has not stopped them. I made changes to my diet inline with a NKF publication at the same time, cutting way down on my beloved almonds and chocolate. I recently had the LithoLink test done and discovered this site, and am implementing this site’s recommendations as they apply to me test results. My oxalate levels are still way too high, but my calcium and other levels are good.
Fortunately my stone events are usually only noticed because of brief periods of hematuria. I have had some uncomfortable episodes, but have not gone to the hospital or had surgery.
I hope this is helpful to someone.
Steve
Hi Steve, Thanks for writing. You are not alone in this practice and I see nothing wrong with it so long as you have a good food scale. The high oxalate should be fixed, no doubt dietary. I would presume urine citrate is normal. I would be sure to check on new stone rate with a CT scan very few years. Radiation dose from modern machines is very low. Regards, Fred Coe