The pretty picture shows uric acid crystals in urine magnified 400x under polarized light to bring out their shapes with special clarity.
Here is what to look for and what to do if your stones have uric acid in them or are entirely uric acid.
A caution: Occasional stones are not uric acid but a salt of uric acid: Sodium hydrogen urate or ammonium hydrogen urate. This article is not for them, They occur in unusual situations, not as yet covered here or anywhere else on this site.
Urine pH
 Uric acid is a large carbon based molecule. Water ‘dissolves’ molecules because of charge attraction. But there is not a lot of charge on this kind of molecule. In fact there are only two sites, both negative and both where nitrogens (N) are. One of them is set up so that it is always blocked by a positively charged proton (H), the other, the nitrogen (N) at the lower right of the molecule can be blocked or not depending on the pH of the urine.
Uric acid is a large carbon based molecule. Water ‘dissolves’ molecules because of charge attraction. But there is not a lot of charge on this kind of molecule. In fact there are only two sites, both negative and both where nitrogens (N) are. One of them is set up so that it is always blocked by a positively charged proton (H), the other, the nitrogen (N) at the lower right of the molecule can be blocked or not depending on the pH of the urine.
At a pH of 5.35 about 1/2 of the uric acid molecules will be blocked and one half will have an open (negatively charged) nitrogen at the lower right. It is this one site to which water molecules can bind to hold the molecule in solution so it does not crystallize and produce stones and gravel.
Each kink where two lines join in this drawing is an atom. Carbons are just kinks, nitrogens hydrogens and oxygens are labelled. So, count them: Nine in the two rings, 3 oxygens (O), four nitrogens (N) and 1 to 2 protons (H) making 16 – 17 atoms with only one charge site.
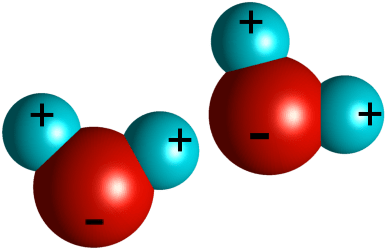 Water molecules have three charges for three atoms. One end of the big red oxygen atom has the two protons (the two H of H2O) bound to it which are positively charged. The other side of the oxygen atom has a negative charge (- sign along the bottom of the big ball). The two water molecules shown here pull each other together because the proton (positive) atoms attract the oxygen atoms (opposites attract), and that is why water is continuous – you can swim through it, pour it, drink it – and cannot pulled it apart.
Water molecules have three charges for three atoms. One end of the big red oxygen atom has the two protons (the two H of H2O) bound to it which are positively charged. The other side of the oxygen atom has a negative charge (- sign along the bottom of the big ball). The two water molecules shown here pull each other together because the proton (positive) atoms attract the oxygen atoms (opposites attract), and that is why water is continuous – you can swim through it, pour it, drink it – and cannot pulled it apart.
Imagine this in three D. Water is innumerable of these molecules holding each other positive to negative sides even though when we drink it we would never know how it was put together. Because there is one charge per atom, there is a lot of force holding these atoms together.
Now, imagine uric acid swimming among these water molecules. The small positive balls, protons, will bind to the negative nitrogen (N) on the big carbon ring, and only that will keep uric acid ‘in solution’, which means the uric acid will be held in among the water molecules as if it were one – though it is too large to really fit in smoothly and has very little charge (1 site for 16 – 17 atoms) compared to water which has 3 charges for three atoms.
The form of uric acid with one charge is called urate, denoting it has a charge and can form a salt pair with another atom or molecule, like calcium does with oxalate or phosphate, or sodium does with chloride. The usual salts are sodium and ammonium urate. One can dissolve hundreds of milligrams of uric acid as urate in a liter of urine.
As you begin blocking the one charged site with protons (1/2 blocked at pH 5.35) less and less can be in solution. When both sites are ‘entirely’ blocked there is only a tiny residual charge and no more than 90 mg of this ‘uric acid’ – ‘acid’ because having both its protons – can be dissolved. The little bit dissolved is because of residual charge shining through or around the blocking protons (I realize my imagery is not exact here, those of you in the business will forgive me).
I have taken some time and space over this detailed matter because it is more or less all anyone needs to know about uric acid stones.
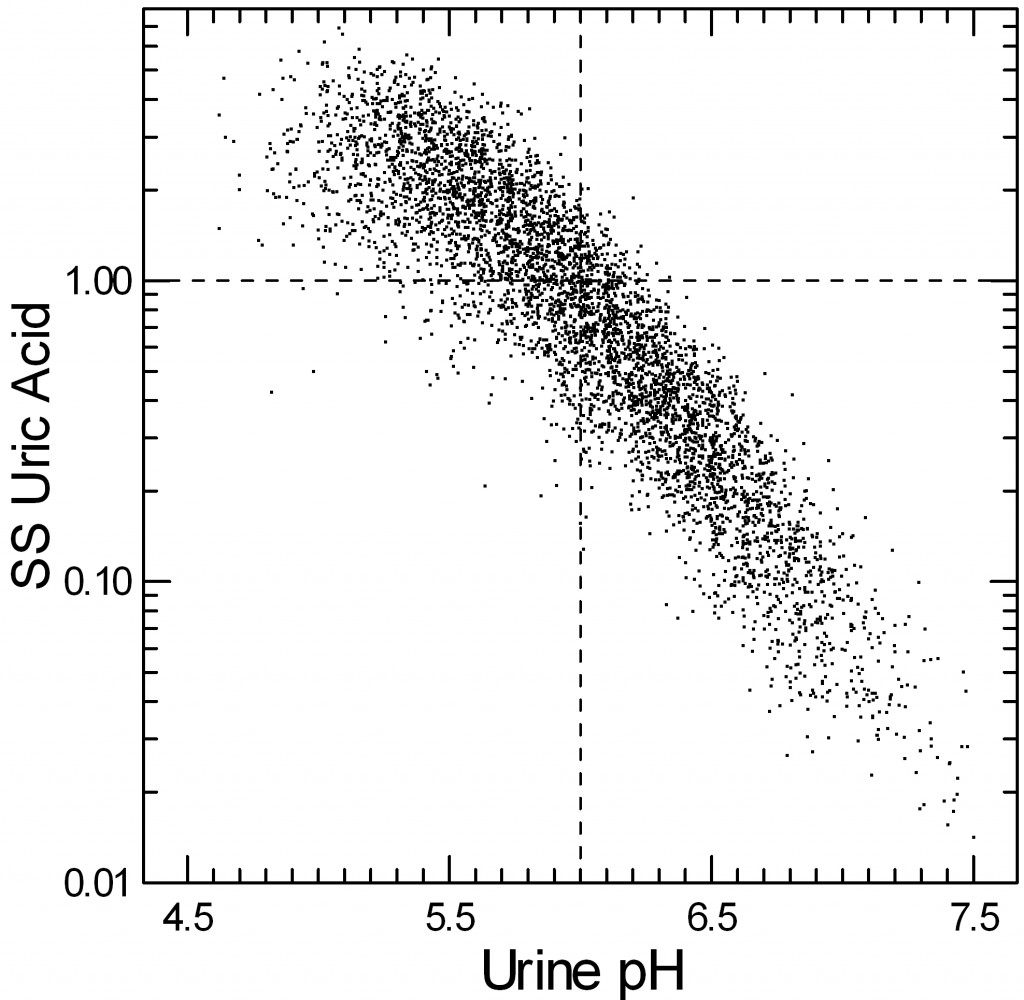
Urine Supersaturation vs. Urine pH
If you do not already know about supersaturation, stop here and read the Five Steps of Stone Prevention. Values above one mean uric acid crystals – and therefore stones – can form and grow. Below one, uric acid crystals cannot form, and those already present can indeed dissolve – albeit slowly.
Each point on this graph comes from a single 24 hour urine collected mostly by me in my stone clinic at University of Chicago. The horizontal dashed line at 1 is the supersaturation demarcator I just referred to – above it is supersaturation, below it is undersaturation.
The vertical dashed line at pH 6 is the perfect place. Below it, supersaturation becomes increasingly common as you can tell by the fraction of tiny points above 1. Above 6 only a tiny fraction of points are above one – in the lower left corner of the upper right quadrant of the graph.
Urine volume, and the total amount of uric acid excreted have an effect – that is why the graph is a thick band and not a thin line, but pH is so overwhelming it takes all of the real clinical focus.
Read Your Report
Uric Acid Supersaturation is Above 1
Is it above 1? Lower it. The ways are simple. Raise urine volume above 2.5 liters daily and raise urine pH just above 6. Recheck in a few weeks, and adjust if necessary. When supersaturation is below 1 and pH about 6 you are done, but be sure and check again at 6 months – habits can be difficult to change long term.
Calcium phosphate stones are fostered by too high a pH, so just above 6 is enough!
Sometimes just very low urine volumes can create a supersaturation for uric acid above 1 even at a urine pH of 6. Though uncommon there are a few points with such supersaturations in the right upper quadrant of the graph above, as I have noted. Simply raise urine volume, recheck, and if supersaturation is now very low and pH still about 6 perhaps that is all you need to do.
Uric Acid Supersaturation is Below 1
Is uric acid supersaturation below 1?
You have a problem.
The urine you are producing could not have supported uric acid stone formation, yet you did indeed make such stones.
This means something has changed from when you made the uric acid crystals and when you collected the urine. One possibility is that you know what it was, the change is permanent, and you have cured yourself.
The other possibility is that you do not know what it was that changed, and you therefore do not know when those risks for uric acid formation will return. Perhaps your physician can prompt you but frankly it is mostly up to you to think about what might have happened. Were you dehydrated a lot of the time? Had your diet been odd? For example popular all protein diets for weight loss might lower urine pH a lot.
How to Increase Urine pH
Assuming you are the usual uric acid stone former you will increase urine volume and pH. The former is higher fluids, and the latter is some form of alkali, usually potassium citrate.
Potassium Citrate
The pills are 10 mEq (units are in the prior article on reading your reports) each. No one can know exactly how much to give at first but a common starting place is 2 twice a day or 40 mEq. The day contains 1,440 minutes and we want the urine pH to be as even as possible for all of them, and not very acidic (below 5.35) for any of them. That is one reason for using two doses a day. An even more ideal dosing would be 3 daily but people tend to miss the middle dose.
Citrate is not an alkali and cannot itself make the urine alkaline. It works because it is metabolized in the liver and kidney in such a way as to create bicarbonate – the buffer of the blood. Sodium or potassium bicarbonate themselves could make urine more alkaline, obviously, but they have a drawback. The sodium salt imposes a sodium load which could raise blood pressure. Either salt will be absorbed rapidly and create a relatively short spike in urine pH compared to citrate which is absorbed and then metabolized – a slower process that gives a more even urine pH result.
Because of regrettable increases in pricing by the few manufacturers of potassium citrate and an unwillingness of insurance companies to pay numerous workarounds have been proposed on this site. But all of these involve use of an alkali so lab changes are much the same.
Other Alkali Sources
One commercial beverage, Crystal Light contains in one liter the same alkali amount as two 10 mEq potassium citrate pills, and it is inexpensive. A common approach is to use it to replace a two tablet dose as desired, and the extra fluid can count toward the day’s needs.
Sodium bicarbonate tablets – 2 10 grain tablets – can be purchased OTC and will more or less make up the alkali of one 10 mEq potassium citrate tablet. The sodium load of 40 – 50 mEq daily is not ideal, and absorption and urine loss are rapid so the treatment is more likely to worsen hypertension and more fleeting.
Lemons and other fruits. There is no doubt that citrus fruits contain citric acid, but the amount of citrate will depend on the amount of the acid that is in the citrate – not the acid form. One half cup of Real Lemon provides just 6 mEq of citrate and has to be dissolved in a lot of water and sweetened. The link shows all of the beverages and juices for which we have measurements. The extensive comments are very valuable and have in them a lot of practical ideas.
Do Another 24 Hour Urine with a Blood
As I mentioned in passing before, one expects certain changes in urine with potassium citrate, so lets look for them.
Urine Potassium
If you take it you should find it in the urine. So if the baseline urine potassium was 40 mEq/24 hours, and you added 40 mEq/24 hours you should expect 80 mEq/24 hours. In general this will be true. But unlike diet sodium that is almost totally absorbed and excreted in the urine, potassium may not be completely absorbed, and some may enter the vast cell potassium stores or even be excreted by the intestine itself.
A good reading of the lab sheet is, nevertheless like this. Urine potassium should rise more or less in proportion to the potassium citrate dose – if there is no rise, the material is not being absorbed.
Urine pH
You should expect a rise in pH and your goal is about a pH of 6. There is no benefit to much higher, and a real risk that too high a pH (above 6.3) will increase supersaturation with respect to calcium phosphate and possible new stones of that mineral.
If the pH has not risen enough, check the urine potassium. Did it rise more or less in line with the dose? If not, perhaps the drug absorption is too low or you missed a dose. If it did rise, then you need more alkali. So, in either event, increase the dose of alkali. A good formula is to go from 2 twice daily to 2 three times daily – longer span of treatment, but do not skip the middle dose!
If the pH has not risen at all, become suspicious.
If the urine potassium did not rise as expected, suspect the tablets are not delivering the drug from their binding envelope. If the urine potassium did go up as expected, the pharmacy probably has substituted potassium chloride for potassium citrate. That is legal as people usually are after the potassium.
Urine pH test strips are a useful day to day checkup especially if stones keep forming. Dips are smoothed out in 24 hour urines. Walmart, Amazon, and CVS all have inexpensive urine pH strips. Shop for price and colors that work for you.
But even with strips as an ad hoc monitor do 24 hour follow up tests because they are your average, and the average is the overall effects on crystals in a day. The two approaches are complementary.
Urine Ammonia and Sulfate
Because kidneys remove acid from the body via ammonia excretion – about 2/3 of the total daily, giving an alkali will lower urine ammonia excretion. But there is a special problem: How do we know if the acid load itself has changed?
The answer is the urine sulfate, and here we are entering into real professional reading of laboratory tests. Sulfate is the final oxidation product of sulfur molecules on cystine and methionine, two amino acids; as it is oxidized this sulfer is converted to sulfuric acid. That is a principal part of the daily acid load. The amounts are perhaps 2 to 4 mEq per kg of body weight but vary with meal choices.
Your 24 hour urine test should have urine sulfate as well as urine ammonia and you use them together to assess the effects of your alkali treatment. Whatever the alkali, citrate, bicarbonate, pills, beverages, juices, ammonia will fall in relation to sulfate.
Lets do an example. Your baseline – before treatment urine sulfate is 50 mEq/24 hours, your ammonia excretion is 40 mEq/24 hours (the ammonia is almost always below the sulfate because the kidney has other ways of removing acid), and the pH is 5. You take 40 mEq of potassium citrate. The next urine shows 60 mEq of sulfate – more protein intake that day, perhaps, and the ammonia is 15 mEq: This is what one expects in a general sense.
You will usually not be able to add up the exact changes in ammonia, dose of citrate, and changes in sulfate as in an accounting lesson: Too many ways the system can respond. But the general sense is that ammonia must fall in relation to sulfate, and in a fairly reasonable approximation to the dose of citrate.
Urine Citrate
Surprisingly, urine citrate might not rise much even though ammonia is suppressed, and urine potassium and pH rise. Citrate handling is very complex, and in this area of disease all we really care about is the pH so that uric acid will not crystallize.
Some people, especially those with diabetes or so called ‘pre-diabetic’ people with insulin resistance may have very high citrate excretions, as high as 1,500 to 2,000 mg/24 hours. When you give potassium citrate, sometimes urine citrate rises so much that a significant portion of the dose is simply lost in the urine and not metabolized to bicarbonate. If so, simply use more.
Urine Uric Acid
I have not even mentioned how much uric acid you excrete daily because it has virtually no influence on whether uric acid stones will form. But it can change in what will appear an odd manner.
Not rarely, when urine pH is low, uric acid is crystallizing out of the urine in the 24 hour urine collection container, or even in you, so the amount in 24 hours will be normal or low. When the pH is increased, the amount will ‘increase’ because no more is crystallizing, and people – even physicians – can wonder why. Why is because none is being lost through crystallization.
Drugs and diets that aim to lower uric acid production and excretion have no role in prevention of uric acid stones. Fluids and alkali are all that matter.



How do I access my “Kidney Stone Lab Report?”
I went to the North Shore Connect website . Under “Test Results” the newest item is 5/04/2015
thanks,
Bob Howard
Hi Bob, Sounds like you found it. Now, use the two articles to read it and see if it makes sense. Regards, Fred Coe
The 5/4/2015 test results were from Highland Park Hospital. Since then, I have had other tests (including a CAT scan). I am under the care of Dr. Zissman and the U of Chicago Hospital. I did a 24 hour urine collection (or 2 or 3 – I forget) and did a blood draw in the Summer/Fall 2015 and I don’t know how to find the test results. I was hoping to see something showing my results compared to the normal range. I suppose I could ask Dr. Zissman, but I assume that she would contact me if there was some issue. So I responded to your articles. Thanks for your help.
Bob
Hi Robert, I guess there are two questions here. I presume that Dr Zisman has or will soon talk to you about your results. The other level is for you to get your own results and look at them on your own. For the former, I would call her directly. For the latter, you should have your results and look at them as the article says- it is not normal ranges but stone risk and the article shows that for calcium, volume, oxalate, citrate and the supersaturations. Let me know here if the article does not do it for you because I will want to know – editing online is easy! Regards, Fred
Hello Dr. Coe,
I certainly understand that drinking more and diluting ones urine decreases supersaturation. My question: When acutely increasing fluid intake over a few days, is there a net change in total daily urinary uric acid excretion until new steady state achieved? Perhaps due to mild hypervolemia decreasing renal urate rebsorbtion?. If so, can you point me toward a reference in the literature? thank you in advance. Jesse
Hi Jesse, Thanks for the comment. If the drinking is water, there will be no effects on uric acid excretion but if the beverages impose a sodium load there may be because proximal reabsorption will fall and URAT – the transporter – can be affected. Water alone will not volume expand in an otherwise normal person as fall in VP will immediately lower CD water reabsorption. Even so the effects of sodium will be small. But, if SS falls and there has been prior crystallization – very common – then the urine uric acid excretion can rise dramatically, essentially to its true level. This is a very deceptive picture because the low volume uric acid SS will be around 1 when crystallization is occurring and may even rise as volume goes up! Regards, Fred
Dr. Coe,
You mention above:
“Below one, uric acid crystals cannot form, and those already present can indeed dissolve – albeit slowly.”
Is there any way to understand the rate of dissolution for Uric Acid stones already present? I have several large Uric Acid stones 12 and 18mm in my left kidney as seen on a CT scan. I have started on Urocit-K and increased my pH above 6. I am curious about how long it may take to dissolve stones of this size. Working with my Urologist, I am scheduled for another CT scan in 3-4 mos. Is this enough time to see a noticeable reduction in size provided I can maintain the higher pH? Is there any benefit to even higher pH – above your recommended 6.3 – to dissolve existing stones?
Thank you in advance.
Hi John, The rate of volume loss is dependent on the size of the stones as loss occurs off of the surface out of the volume, and the surface/volume ratio is ~1/r. Raising the pH much above 6.3 is not wise as calcium phosphate crystals can be promoted and at pH 6.3 you are 1 pH unit above the uric acid pHa and SS UA will below 1. The stones are almost certainly not singular but made up of smaller stones glued together, so the 18 mm one will probably begin to break up into smaller stones. Message: wait, leave the pH where it is. Regards, Fred Coe
Thank you Dr. Coe! I really appreciate your response. This site is extremely helpful for patients dealing with stones. Your no-nonsense practical approach to dealing with this wonderful. I am glad I happened upon your site this morning. Have a great weekend!
Hi Dr. Coe,
Could you please try to help me understand the implications of “the ammonia is almost always below the sulfate because the kidney has other ways of removing acid”.
Say NH4 24 was initially around 25 and Sul 24 around 50. Then after starting Chlorthalidone 25 mg, (no potassium citrate yet and fairly constant diet), both NH4 and Sul24 were around 50. And this pattern has persisted for a year even with various doses of potassium citrate from 5 to 40 mEq. NH4 went even higher at times with no infection or other known cause.
Any idea why this might happen and whether it provides any clues as to how the body is or isn’t handling acid excretion normally?
Appreciate your time. Best regards,
Al
Hi Al, ammonia will normally be below sulfate for the reason you mention. Chlorthalidone invariably causes some potassium loss from cells – serum normal – and the kidney responds with increased ammonia. Take a look here. Regards, Fred
Hi Dr. Coe,
Thanks very much for your reply. That is a familiar article. A challenging, but rewarding read!
Your hypothesis makes sense, so let’s test it with the most trusted data from the last year:
Chlorthalidone/Potassium Citrate -> blood potassium, NH4 24, Sul 24
0mg/0mEq -> K=4.0, NH4=29, Sul=48 (baseline)
25mg/0mEq -> K=3.8, NH4=59, Sul=59
50mg/40mEq -> K=3.8, NH4=52, Sul=53
25mg/5mEq -> K=3.7, NH4=61, Sul=49
Wow, does it make sense that the slope of the curve could really be that steep? I didn’t realize a change of 0.2 in potassium could be so significant.
Take care,
Al
Hi Al, Most potassium is in cells, so when serum falls a very little bit it usually means cells have lost appreciable potassium. The cells know that and kidney cells respond with – among other things – a rise in ammonia production. pH inside the cells fall. Nice observations. Regards, Fred
Would not allopurinol help lower uric acid production and be helpful in those whose liver produces too much?
Hi David, No; uric acid stones arise from low urine pH and allopurinol is not necessary or sufficient. Regards, Fred Coe
Hi Dr Coe,
An ultrasound shows 13mm stone. This current stone has formed since lithotripsy (x 2) to remove a 3cm calcium oxalate stone a year ago. It was assumed to be the same sort until it was not seen in a recent X-ray. I have been maintaining an increased fluid intake and about 3/4cup of fresh lemon juice daily for Several months before current ston was discovered as a preventive measure. Recent fasting blood and 24 hour urine show volumes just over 4l they do not show ph or supersaturations though everything else is within normal range –
9/1/17
Creatinine 6.9 mol + citrate 3.8 mmol + oxalate 0.32 mmol)
19/1/17
creatinine 11mmol + calciul 5.3
6/2/17
creatinine 10+ Urate 2 mmol
Could it still be a uric acid stone?
Thanks for reading,
Anna
Hi Anna, Perhaps the 4 liters of volume is not always present – perhaps during the day it varies too much. Your calcium is high – 5.3 mmol (>200 mg/day) poses risk. Without a urine pH one can say nothing about uric acid, but your stone was calcium oxalate and the new one is likely to be the same. Here is the problem, the 24 hour studies are fragmented and incomplete – no supersaturations, no pH, no sodium; can your physicians obtain better for you? If not the only approach is to assume calcium oxalate and pursue prevention via diet and if needed medications. I presume you have no known diseases causing the stones. Regards, Fred Coe
Thanks for responding – I will follow up with my Gp regarding supersaturations, sodium & ph – I was hopeful it would not be calcium oxalate as it didn’t show on a plain X-ray – but I do understand that different types of stones can coexist and I imagine that can leave room for doubt? from what you’ve shared here it seems I need to focus mainly on calcium oxalate prevention and not worry about ph unless it is flagged as an issue in any further testing. I’m very grateful for your input Dr Coe.
Kind regards,
Anna
Hi Dr. Coe,
Very interesting article. When trying to identify uric acid stones (vs non uric acid) in the kidney are you familiar with Dual Energy CT scans? Is this technique widely available and is it accurate in predicting uric acid stones? Please see link below for reference.
http://www.scbtmr.org/Portals/9/Meetings/2012/Final%20stone%20Dx%20and%20Mt%20SCBT%202010.pdf
Thanks
Jason
Hi Jason, The link you sent dead ends so I cannot read the paper. I am not aware of dual energy CT for this purpose but for uric acid routine CT is not too bad because the density of uric acid stones is lower than for the others. Regards, Fred Coe
Hi Dr. Coe.
Sorry the link didn’t work. Here is another link.
Since you said routine CT is not too bad, below what HU density would you consider it to be uric acid stones?
http://www.massgeneral.org/imaging/news/radrounds/july_2008/
Regards
Jason
Hi Jason, Thanks. I gather the dual scanner is good for uric acid but not so good for cystine, struvite and mixed stones. As such, perhaps, it is good for stones with low HU that might reflect poor centering or small size. HU below 500 raise suspicion of uric acid, especially when stones are large. I did not see the additional radiation dose or cost estimates but one would have to weight the modest improvement against both. Very interesting. Regards, Fred
Thanks Dr. Coe,
What is the size demarcation between a large stone and small stone?
Regards
Jason
Hi Jason, It depends. If we are speaking about passing, probability begins to go down at 5 mm and becomes low above 7 mm. If we are talking about large enough to need percutaneous nephrolithotomy and contra-indicate SWL it is 20 mm. Regards, Fred Coe
Hi Dr. Coe,
I think you misunderstood. I meant it in the context of your answer to the previous question. You said “HU below 500 raise suspicion of uric acid, especially when stones are large”. What size of stone would you consider large as opposed to small, where HU is not reliable?
Regards
Jason
Hi Jason, Stones of less than 2 – 3 mm are hard to image, so large enough for HU is perhaps 4 mm, depending on the quality of the CT. Regards, Fred Coe
Hi Dr. Coe,
With regard to your Uric Acid SS vs pH graph, I think I can understand that if I increase my urine pH, my SS can be lowered below 1. It doesn’t work in the reverse, correct? i.e. if I lower my SS (by drinking lots of water), does my urine pH increase correspondingly?
Also how much does the pH of the water I drink have an impact on my urine pH? Can high pH water be used to dissolve uric acid stones instead of K citrate?
Best Regards
Jason
Hi Jason, I want to be sure I am not doing you a disservice. I am answering detached questions for you but perhaps they might mislead your care. Do you in fact have uric acid stones? Do you have a physician to care for you? Taking on diet or other factors to alter urine pH is not always good. Higher urine volumes are, however. Perhaps you might clarify matters a bit more for me so I am sure I am doing no harm. Regards, Fred Coe
Hi Dr Coe,
Sorry I shouldn’t have written in the first person. My uncle has had uric acid stones, I was trying to explain to him the science behind it. Maybe you can explain just the graph, since I don’t know all the details of his case.
In the Uric acid SS vs pH graph, if one decrease the SS below 1 (by drinking lots of water), does that automatically increase the pH level of the urine? or only the converse is true (i.e. by increasing pH level, the SS will drop below 1).
Thanks
Jason
Hi Jason, Only change of pH matters. The graph shows SS vs. pH; there is no water or urine volume on the graph. Water will not do it. Regards, Fred Coe
I recently was diagnosed with 6 kidney stones in my right kidney (the largest are two 7mm stones) and multiple small stones in my left kidney. I discovered the issue while I was pregnant and began to notice small specs of blood in my urine. The odd thing is that I go annually to have my kidneys and gallbladder scanned with an ultrasound due to my gallbladder polyp. For the past several years they have detected only 1 stone in each kidney. When I asked my urologist why I suddenly have so many stones, he claimed that I couldn’t have grown all of those that quickly and they must have been missed in previous ultrasounds. My thinking was that maybe the prenatal vitamins that I have been taking for several years while trying to conceive caused the rapid stone growth, but my urologist just seems to think they have been there for a while. I was treated for asymptomatic bacteria in my urine twice while pregnant. My urine cultures while pregnant showed white blood cells, blood, amorphous crystals, and leukocyte esterase. I have passed 3 stones before but have not managed to catch any. My most recent stone was passed a couple of months after having my baby. They detected the 5mm stone in my right ureter and I strained my urine but never caught the stone. I went in for a repeat ultrasound to see if it passed and they did not see it in my ureter anymore. My 24 hour urine test showed low citrate and high uric acid levels. I have a high urine volume (4 liters). Can I deduce what types of stones I have from this information? My urologist told me to go on a low uric acid diet and to drink potassium citrate. Is that what you would recommend as well? I’m worried that he isn’t being proactive enough with his approach. Can you offer any advice on treatment? Thank you!
Hi Katie, I understand and suggest you do indeed need a full evaluation. Here is a good list of steps. See if they have been followed. Above all do not leap into treatment without a full evaluation. Regards, Fred Coe
Hello Dr. Coe,
I would like to know how long would it takes to dissolve a 0.5×0.2×0.5cm and 0.4×0.2×0.4cm (200 to 400 HU) KIDNEY STONE? im taking tascit at 15mEq twice a day and 2tab of sodium bicarbonate 650mg 3x a day.. im taking medication fr almost 4months and the stone was stil there with my last sonogram result it was noted that there is no significant interval change in size.
How long would it takes to dissolve kidney stones ?
Hi Joefferson, I do not know. Firstly the stone may be mixed uric acid and calcium salts. Possibly it is all calcium and simply HU does not discriminate. Sodium bicarbonate raises urine calcium so if it has calcium salts things might be less than ideal. Was your urine pH low before treatment? Do you have other reasons to believe your stone is uric acid? Let me know, Regards, Fred Coe
Hi Dr. Coe,
Would you be able to recommend a doctor or two in the Detroit area?
Thank you,
Hi Steve, I gather it is for kidney stones, but you do not indicate if for surgery or medical prevention. They go together only rarely. Please let me know which and I will try. Regards, Fred Coe
Hi Dr. Coe,
Thank you for the article. It has been helpful in more ways then one. I know you’ve written this a while back but I do hope for a response.
I was wondering how drinking citric acid may help?
I was prescribed potassium citrate but it made me feel quite ill. I’ve read a lot up on lemons and crystal light for urine alkalization. I’m not terribly clear on how drinking these help raise the level on urine ph but the common denominator in the potassium citrate, lemons and crystal light is citric acid. They sell online food grade citric acid. It’s a white powder, very acidic, used mainly to coat candy to make sour candy. I was wondering if consuming small but significant enough amounts of the powder (I would consume with water, I tend to love sour drinks and food so no added sugar).
I will link the package below. If this citric acid will function the same as aforementioned sources in alkalizing the urine, can you suggest how much powder would be effective enough while remaining safe as consuming that much citrate can be hard on the stomach. Thank you!
Milliard 100% Pure Food Grade Citric Acid – Non-GMO 4.54 kg (10 Pounds) https://www.amazon.ca/dp/B00GNBHPAS/ref=cm_sw_r_cp_api_i_iNJ5EbG4FJQCT
Hi Rihanna, what causes increase in urine citrate, and helps prevent stones, is the citrate ion. It is metabolized as citric acid, and in so doing takes up a proton from the blood buffers thereby creating bicarbonate alkali. Citric acid is useless as it is merely metabolized – if at all – as the acid, or excreted unchanged in the urine. So the common denominator is citrate ion – with whatever cationic counter ion you can get such as potassium, sodium, or calcium. Regards, Fred Coe
Hi Dr Coe,
Thanks for the fantastic article.
Regarding uric acid stones; the common denominator seems to be citrate. If one were to consume purely citric acid (powered form, which is usually used to coat candies to make them sour), would that be effective in alkalizing the urine?
And how much would be necessary daily to have an actual effect? Are urine dipsticks a good way to keep track or the at home ones tend to be faulty?
Thank you! Be well.
Hi Rihanna, I already answered about citric acid in your prior query. Usual doses of alkali are 40 – 60 mEq in 2 – 3 divided dones. One needs 24 hour urine testing as the daily average pH gives a better sense of average uric acid supersaturation. If you want to use a dipstick, collect the whole urine and test its pH. Regards, Fred Coe
Hello,
Do you an entry where you’ve written on low potassium and low magnesium in regards to oxalate/calcium stones? My urine pH was also high around 7.6
Thank you.
Hi Lyndsea, Not exactly, as these are due to underlying problems, not of themselves. If you can provide more information I will try to help. As these are important in themselves, please feel free to write to me. Regards, Fred Coe
Hi Dr Coe,
I have high uric acid which I control using Allopurinol 300mg/day and I had uric acid stone as well in the past. Just in recently, I’m able to find a doctor who wants to prescribing Potassium Citrate to increase my urine pH to prevent uric acid stone.
My questions:
1. I think I read somewhere on your blog that at minimum, urine pH has to be 5.5. If this is correct, my question: does maintaining urine pH at 5.5 at the minimum applicable to everyone and in all situation? FYI, prior to consuming potassium citrate which I started in Aug of this year, my urine pH was always at 5.
Or, does how high the urine pH should be, will be determine by the 24 hours urine uric acid? Meaning, the higher the 24 hours urine uric acid, the higher the pH should be?
FYI, my most recent 24 hours urine uric acid is 4.6 mmol/day, would maintaining urine pH at 5.5 good enough in my situation? Or, do I need to maintaining a higher pH? If so, is there a guideline that I can use? For example, if 24 hours urine uric acid is between 4.6 – 5, then I have to maintain pH at 6.5 or higher, if 24 hours urine uric acid is between 4 – 4.5, then I have to maintain pH at 6 or higher.
2. If urine pH is related to urine uric acid, does it have to be 24 hours urine uric acid? What if I use on the spot/random time urine uric acid?
3. Does consuming orange juice can increase urine pH? If so, how much of an orange juice needed to increase the pH? Would consuming 2 eight ounces glasses of orange juice daily enough?
4. I had 2 episodes of uric acid stones in the past and whenever I had the stones, my renal function was also lower than normal. I have yet looking at my pH at both episodes.
In July, my renal function was also lower than normal. Creatinine = 137 umol/L, eGFR = 53 mL/min/1.73m2, uric acid = 515 umol/L.
In Aug creatinine = 135 umol/L, eGFR = 54 mL/min/1.73m2.
In Sept creatinine = 99 umol/L, eGFR = 78 mL/min/1.73m2, uric acid = 397 umol/L
In Sept, I did ultrasoung and they found no stones.
In Aug, I started consuming Potassium Citrate and monitor my pH every day.
I’m wondering however, the number significantly jumping from Aug to Sep and wondering if there is anything that we can make out of it?
Thanks Dr. Coe!!
Hi Niko, uric acid solubility is so dependent on urine pH that nothing else much matters. At pH 5.35 50% of uric acid is in the diprotonated state, which has a solubility of only 90 mg/l. At pH 6 almost all uric acid is monoprotonated and one can dissolve over 1000 mg/l. The goal is a urine pH of 6 on a 24 hour urine, the latter because your average over the 24 hours. Spot urines are just that, and leave room for crystals to form. If pH is on average 6 or more net crystal formation will cease – and stones. Orange juice is never enough, potassium citrate is the ideal, about 30 – 40 mEq/d in divided doses. That is essentially a cure. As for changing renal function, uric acid can form extensive crystal masses – not fully formed stones – and temporarily reduce kidney function. That could be a reason for you. With the treatment noted such falls in eGFR should stop. Regards, Fred Coe
Hi Dr Coe,
Just want to make sure that my understanding is correct.
1. How much uric acid in the urine has no role in determining how much pH I should maintain, correct? The goal is urine pH of 6 on a 24 hours urine, regardless of the amount of uric acid in the urine, correct?
2. I think I read at one of your article which says that urine pH that is too high is also not good and may create kidney problem? Somewhere between 6 to 6.5 is the ideal?
Thanks Dr Coe!
Hi Nick, yes. A 24 hour urine pH around 6 – 6.5 is ideal, and within that range no reasonably achievable amount of urine uric acid will lead to uric acid stones. Regards, Fred Coe
Hi Dr Coe,
So, I’ve been doing trial and error with regards to the potassium citrate dosage. I’m using urine pH strip test and I test my urine in the morning before I had my breakfast.
In the beginning, I took 20mL a day (10mL in the morning, 10mL in the evening). My urine pH was between 6-6.5. Not sure why I did what I did, but, I tried to took 30mL a day and sure enough, my pH was around 7.
Interestingly, when I lowered the dose to 10mL a day, my pH was even higher at around 7.5. Any reason why this is happening? Or this is simply a strip test not being accurate?
Hi Nick, Uric acid stones respond to the daily average urine pH. You are sampling here and there and that is confusion. For example, morning urine pH before breakfast is often high for reasons I know but are not important here. Given the horrid nature of UA stones and that raising 24 hour urine pH prevents them I would advise 24 hour testing as my guide. Regards, Fred Coe
Hi Dr Coe,
I consistently test my urine in the morning before breakfast so it is not really “here and there”, however, I understand that 24 hour urine pH is the objective and ultimate way of measuring the pH for UA stone prevention.
The problem is, where I live (Alberta, Canada) increasing pH as UA stone prevention is not common. I consider myself lucky when I finally found a doctor who is willing to prescribe potassium citrate but even the pharmacist do not know if potassium citrate is, among other thing, to increase pH.
My doctor also do not give any guideline as to what pH should be the goal, much less ordering 24 hour urine pH test. I have very little faith that I can have my doctor to issue lab requisition for 24 hour urine pH.
That said, if I do the testing on my own by collecting my urine for 24 hour and then use my urine pH test strip, would the result be comparable to the lab testing?
Hi Nick, Given pH is the main issue, collect your own 24 hour urine and measure. It is a more robust estimate of average uric acid crystallization potential. Your goal is above 6. Ideally not too high – between 6 and 6.5. Fred
Hi Dr.. I am a kidney transplant recepient going on 11 years. Suddenly I got knee gout (verified by aspiration). Never had gout in my life through worse kidney function in the past. I injured my knee then the flares started and only then. I am starting potassium bicarbonate to get rid of crystals. Should I add citrate also? My random urine Ph recently was 6. Please advise
Hi Mike, This is complicated. Gout is crystallization of sodium hydrogen urate in joint spaces – like the knee – and unrelated to urine pH. Uric acid stones are made of uric acid itself and more or less cured by raising urine pH to 6. The two are related in that gout often arises because of increased serum urate from reduced kidney function – as can happen with a transplanted kidney. Then, one wants to lower serum urate with medication and potassium citrate is not that medication. In fact alkali will not help with gout. The crystals in the joint need to be looked at because they can be calcium pyrophosphate, not sodium urate, and treatment is different, so be sure your physician has looked at joint fluid aspirate to assure which crystal is present. The latter is more common with joint injury. Regards, Fred Coe
Dr. Coe,
Under Other Alkali Souces, you said that 1 liter of Crystal Light has the same alkali amount as two 10 mEq potassium citrate pills. One packet of Crystal Light “Lemonade” lists 130 mg of potassium & Potassium Citrate is the second ingredient listed after Citric Acid. The Fruit Punch packet and several other flavors first ingredient is Citric Acid but Potassium Citrate is the sixth ingredient listed and Potassium is not listed on the nutritional label at all.
So wondering if the equivalency to Rx Potassium Citrate applies to all Crystal Light options or perhaps “only” to the Lemonade option, if that happened to be the one used in testing? I haven’t looked at them all but haven’t found another of their options, so far, that list a value for potassium.
Thank you for ALL you’ve done and continue to do in educating us “stoners” and the medical profession. I wish I’d found you and Jill Harris years ago. Perhaps I could have avoided 3 hospitalizations since 2019 for Sepsis from my kidney stones, as well as, earlier stone events,
Thank you again.
Hi Carol, It is indeed the lemonade. I just wrote about a lot more K citrate substitutes. Take a look, and be sure and read what I said about their use – or not. Regards, Fred Coe
Dr. Coe,
I submitted a comment/question earlier today about Crystal Light equivalency to potassium citrate and just found you answered it in your article on the cost of potassium citrate. The research was done using the original Crystal Light Lemonade and does not necessarily apply to other Crystal Light flavors.
What do you think of Moonstone as an option?
Just looking for options that I can use to increase my water intake which is a struggle.
Thank you.
Hi Carol, I just sent the link to my alternative alkali article. It is what you are asking for, Fred
Hi … Im learning from your writeup here how to read my 24hr urine lab and seeing if my otc dose is optimal so thanks for this writeup. My health conditions are complicated and I have several main drs but recent labs found Im starting to make urine crystals, im low in potassium and citrate both blood and urine, always have a low urine ph and so after talking with my drs i started taking some over the counter potassium citrate powder i found online and putting a qtr teaspoon in my morning coffee and nightly tea. Wow, what a game changer for the better for me! But its effects now have me wondering some things that neither my rheumatologist or endocrinologist or primary could answer because some of my lab results are a bit conflicting. . How would I go about getting an evaluation and with whom could ask further questions because i believes theres interactions as the potassium citrate is not only eliminating urine crystals but effecting my insulin sensitivity for the better and ive had to reduce my insulin dose which also a good thing for a type 1:) and my insulin sensitivity vastly improved. With the overlap of my many. autoimmune conditions all of which can have kidney complications im not even sure where to consult for questions . The good thing is my kidney function is good, my type 1 diabetes is under control, my sle/ra is troublesome but i want to keep my complications down so im being proactive. I stumbled on several causes of low potassium and citrate that are concerning like certain kinds of dtra and are the source of my questions. Am better off consulting with a nephrologist or urologist?
Hi Staci Ann, I cannot offer much immediate help but potassium depletion does lower urine citrate and worsen glucose tolerance. SLE can cause a potassium wasting problem in the kidney, perhaps a reason for the problem you have in the first place. All I can offer is a telehealth visit, if your insurance includes the university I work for. Alternatively, if you say where you live I can try to identify a university program that might be more convenient. In case the former is practical my secretary is at 773 702 1475. Best, Fred Coe
Thank you – just seeing your reply and i reached out to your secretary phone number for a an appt. I know my more recent labs will likely make sense to you. 🙂
Have you done a study of urine ph vs. blood uric acid. I would like to see this in graphical form if you have along with a correlation if there is one.
Thank you.
Hi Dr Pound, NO I have not made such a graphic display. As you suspect, I suspect the relationship will be inverse because high uric acid in blood correlates with uric acid stones that require a low urine pH, and also with age and reduced renal function which both lower urine pH. Best, Fred