
 Medullary sponge kidney (MSK) is more spoken about than witnessed, and more witnessed than accurately diagnosed.
Medullary sponge kidney (MSK) is more spoken about than witnessed, and more witnessed than accurately diagnosed.
This patient adds to the 12 we have described in our publication, and adds also in having a very long and evolving history with one of us (FLC).
We write for a general audience yet hope to include a level of detail that satisfies physicians and scientists. Here, we may fail of clarity to the one audience or of a sufficiency to the other because the disease is complex. But withal, the evolution of diagnosis and care for this person so educates and the surgical anatomy and histopathology so instructs we have chosen to share the experience.
What is it we are sharing?
MSK is a Unique Disease
MSK is remarkably specific in its anatomy; nothing else is really like it. The terminal nephron segments are tremendously dilated and often filled with innumerable round tiny stone particles. The papillary shapes are distorted by these filled chambers so they become round instead of conical. The masses of tiny stone particles give a radiographic picture that can resemble large collections of stones or calcified papillae, but are neither. Renal tubular acidosis, or just ‘nephrocalcinosis’ come to mind and appear on radiology reports, but neither is the right answer.
The tissue itself is specific to an exactness. The massively dilated papillary collecting ducts are lined by multiple layers of cells, whereas all other people have tubules lined by a single layer of cells. The cells in the interstitium – the spaces between the tubules and vessels – look like those found in embryonic kidneys. The scale of the duct dilation is so great one loses perspective.
MSK has Few Unique Clinical Traits
But for all that, patients with MSK present to even the most discerning clinical eye almost no distinctive traits. They can look like any calcium phosphate stone former can look – papillary calcifications, perhaps multiple stones retained in the kidneys, and little else. When intravenous pyelograms (IVP) were popular, one could diagnose MSK if the contrast agent tracked into the dilated ducts and overlaid the stones there. But obstruction from a stone can create the same ‘pyelotubular backflow’, or can seem to, so diagnosis was fraught. Almost no one does IVPs anymore, because CT scans without contrast suffice for most patient care.
Renal tubular acidosis can come to mind: Masses of renal crystals seem to provoke the idea for physicians even when the blood is normal. ‘Incomplete’ renal tubular acidosis was a name coined to describe patients who formed calcium phosphate stones and had both normal blood and a more alkaline urine pH than 6.5 that did not always fall as much as one might expect with acid loads. In 1992 this patient was so named and treated by FLC who would never use the term today, and by his fellow at the time who is now a world famous kidney stone expert.
Perhaps the most reliable clue to MSK before surgery is done, one we only recently understood, is the combination of a kind of ‘nephrocalcinosis’ with calcium oxalate stones in the absence of remarkable hyperoxaluria.
Nephrocalcinosis is a worn out term, as vague in diagnosis as it is rich in associations. It can mean small calcifications that involve all or most of the papillae, or massive calcifications that fill whole calyces. Frequently one sees the latter, masses of crystal, with calcium phosphate stones, or uric acid or struvite stones. By contrast, calcium oxalate stones usually create the pattern of multiple, bright, small calcifications on individual papillae. It is marked hyperoxaluria that can produce masses of calcium oxalate crystals.
The reason we find massive calcified deposits associated with calcium oxalate stones in MSK is that the tiny proto stones are calcium oxalate yet can bundle in huge numbers within the dilated chambers of the terminal ducts. FLC and his now famous fellow missed the diagnosis despite masses of crystal by radiograph and calcium oxalate stones because neither knew what we know today. The marginal IVP changes of MSK were dismissed because they occurred on the obstructed side.
Pronouns
We are writing this, but separately. FLC is the physician so he writes as ‘I’. AE is the anatomist, and he writes his sections as ‘I’. ‘We’ is reserved for the amalgam of the clinical and anatomical.
The Initial Evaluation
I saw him about about 24 years ago when he was about 25 (real ages and personal details are blurred but accurate enough). He was healthy apart from a recent kidney stone passage, and had been referred to me by a friend. The stone was 65% CaOx and 35% calcium phosphate as hydroxyapatite – no brushite. Because the stone analysis lab used optical microscopy we learned that CaP formed the inner layers of the stone and CaOx grew over it.
An IVP showed ‘bilateral nephrocalcinosis’, but the left side was the more prominent. That side showed marginal evidence of medullary sponge kidney but, being obstructed by the passing stone, prominence of the terminal collecting ducts could have been secondary to the obstruction.
His family members had no stones. He worked at a high intensity job in the entertainment industry with little time to eat or drink. Accordingly there was no breakfast, fast food for lunch, but a reasonable supper with his wife. His physical examination was normal.
Three fasting serums taken between 7 and 9 am showed normal results (not shown) including serum chloride and total CO2. Serum potassium was 4.3.
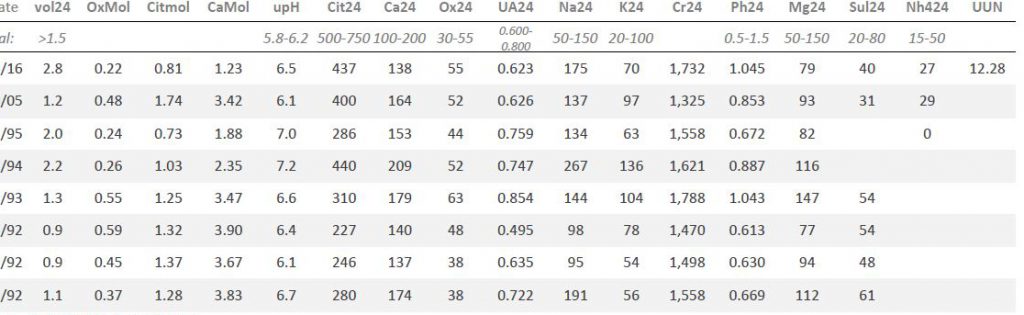
Here is my table of his lab results.
The first three rows up from the bottom (1992) were done before any treatment. Dates are partly cut off for privacy.
Urine creatinine values (Cr24) were all about the same, so the collections seem reliable.
Main urine risk factors: Volume (vol24) and citrate (Cit24) were low enough and oxalate (Ox24) high enough to confer risk. His urine calcium (Ca24) varied with urine sodium (Na24) as expected but at even the highest sodium urine calcium posed little risk. Urine uric acid, potassium, phosphate, magnesium and sulfate (UA, K, Ph, Mg and Sul, respectively) were not abnormal. We did not measure urine ammonia (NH4) or urea nitrogen (UUN) at that time.
Technically, for those who are interested, the molarity of citrate (Citmol) was far below that of calcium (Camol), so more calcium was available for crystallization, and less citrate for inhibition. Urine pH (upH) was variable and thought a bit high.
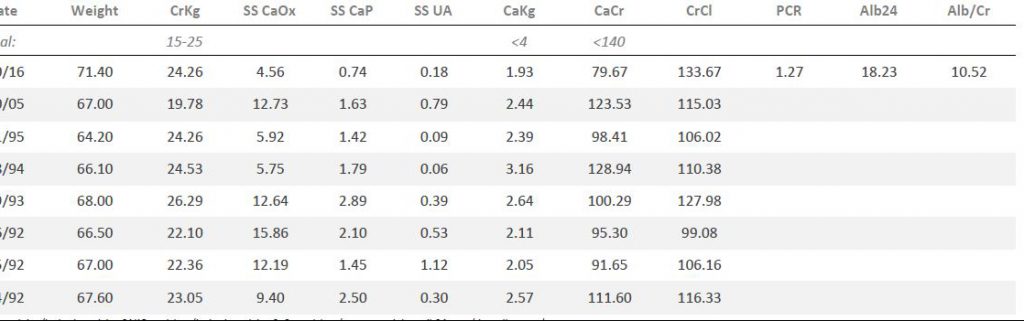
His urine creatinine was high per kg body weight (CrKg) signifying low body fat – expected for a young active man. SS for CaP was higher than usual among normal people, that for CaOx was also impressive. SS for uric acid was below 1 because the urine pH was relatively alkaline.
So, long ago, I cared for a man with a calcium oxalate / calcium phosphate stone and nephrocalcinosis whose laboratory studies showed mainly low urine volume, low urine citrate, and modestly increased urine oxalate, and whose IVP showed what might have been MSK or simply the effects of obstruction from a stone passing.
I, and my renal fellow – now a distinguished professor and director of a program – decided that the low urine citrate along with modestly high urine pH arose from renal tubular acidosis (RTA) – despite normal blood findings, and also that MSK might be the cause of RTA. We treated him accordingly, with potassium citrate 25 mEq twice daily, and fluids, and planned a follow up shortly to see if citrate would rise and CaP SS not rise overly. Likewise we hoped for a new IVP after the obstruction resolved so as to see if the ‘MSK’ was real or merely an artifact.
Looking back, I see higher urine oxalate excretion rates that I would have liked to treat, and suspect it was due to a low calcium diet. Today I would never have accepted the idea of RTA.
First Follow up Visit
In 1993, his urine volume remained low (look back at the two lab tables), citrate had not risen, but urine potassium was higher and serum potassium was 5.07 so he took the medication. Supersaturations with respect to CaOx and CaP were not at all improved. I increased the potassium citrate to 25 mEq 3 times a day and insisted on a new serum potassium a few weeks later – which was unchanged.
Laboratory Follow up Measurements
In 1994 (lab tables above) his serum potassium was 4.5, urine potassium was higher at 136 mEq/d, and urine citrate and volume higher. SS for CaOx and CaP had fallen by half or more, and I felt satisfied. There were no reported new stones since 1992.
In 1995 his serum potassium was 4.4, urine citrate and potassium had fallen, and I realized he had stopped the medication. I strongly suggested he continue it and get a new kidney radiograph to follow any changes.
I heard no more from him for ten years.
Second Follow up Visit
In 2005 I got word he was passing many more stones, and referred him to my urological colleague for management. Serum values were unchanged, urine volume was low; urine citrate and potassium were back to levels seen in 1993 even though he was not taking the potassium citrate. Urine pH was no longer noticeably high. SS with respect to CaOx was very high as was that for CaP – though this latter was below his peak in 1992 and 1993.
I saw him clinically in 2006 after the 10 year hiatus, and he explained he had stopped all treatment in 1995 as I had guessed. He had many stones in his possession and planned to send them to me for analysis. I re-instituted the potassium citrate and more fluids. My records do not indicate if he saw a urologist or what may have happened.
I heard no more from him for 10 more years. The stones were never sent, never analysed.
Third and Fourth Follow up Visits
Third Visit
In the spring of 2016 he came back because of more stones. During the 10 year interval he had passed no stones – which hiatus he ascribed to more fluids – but recently had begun to pass them again.
First there was a new stone, then up to 10 or 12 more. Several of these were analysed. One was 100% CaOx the other 88% CaOx, 12% HA. His serum bicarbonate and chloride were normal, as was potassium (not shown). Urine (lab table is above) showed high volume, and the best citrate ever; the low urine calcium and high oxalate were ascribed 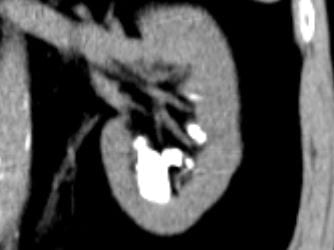 to excess vitamin C and high oxalate diet along with a rather low calcium intake; SS with respect to CaOx
to excess vitamin C and high oxalate diet along with a rather low calcium intake; SS with respect to CaOx 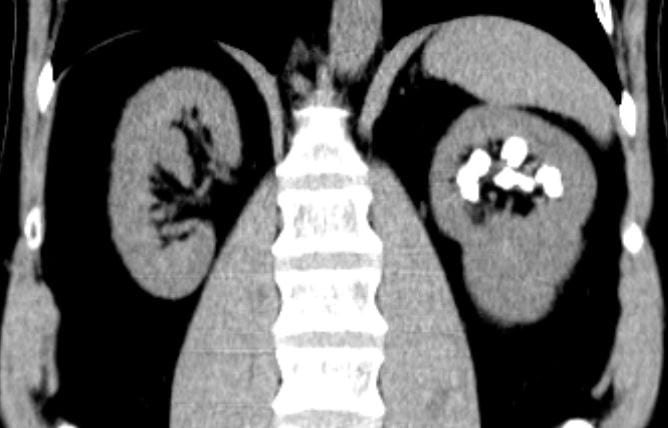 was much lower at 4.5 and for CaP was below 1 mainly from the high urine volume.
was much lower at 4.5 and for CaP was below 1 mainly from the high urine volume.
Urine ammonium (NH424) and sulfate (SUL24) both in 2005 and 2016 were unremarkable. In ‘incomplete’ RTA the former is thought to be higher than normal in relation to net acid load – the sulfate. I did not comment about this in my records as the idea of RTA was no longer one I would consider.
I suggested a lower diet oxalate, continued fluids, and left out the citrate given the history of no stones for 10 years without it. I ordered a new CT.
I believed after this visit that perhaps his new spate of stones was provoked by vitamin C loading – from ‘Airborne‘ and 1000 additional units of vitamin C daily. The Airborne products contain about 1000 units of vitamin C in each pill, so with his supplement the amounts could have been quite large.
Fourth Visit
I saw him again after the CT was available.
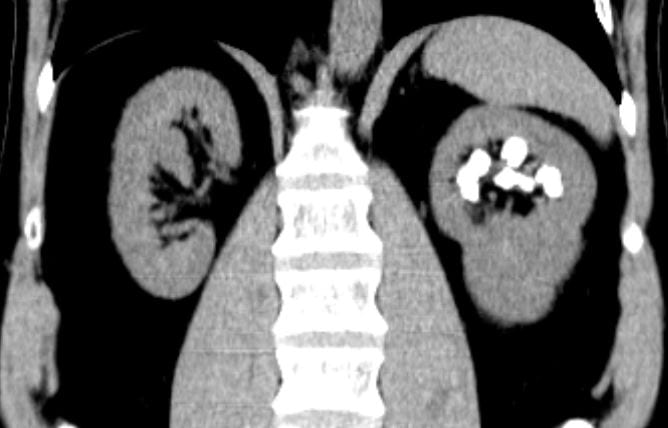
Calcifications were only left sided but involved both the upper and lower poles. The deposits were large, and I could not tell if they were stones, calcifications in kidney tissue, or both.
Transverse views confirmed the disease limited to the left side.
Review of my drawings from 1992 showed calcifications on both sides, which was puzzling. The actual radiographs are no 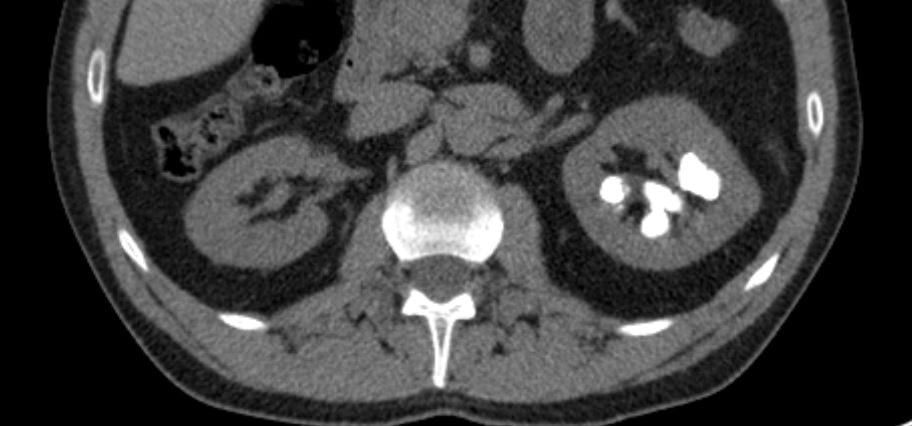 longer available.
longer available.
I was puzzled, and still am, about the conflict between my old drawing and the obvious unilateral stones, but had become convinced the present stones needed to be removed. They seemed large enough and so positioned as to obstruct flow out of calyces and gradually cause injury. Obviously they would never pass.
Lithotripsy was not a consideration nor ureteroscopy – too large. This left percutaneous nephrolithotomy (PERC) that has the advantage of offering – in potential at least – a stone free kidney, and I chose that course assuming my surgical colleague would agree.
As for his stones, I now suspected they arose from a hectic and complex work life with poor hydration and high urine oxalate. His urine oxalate had been hefty all along and there was the recent vitamin C loading. Even so, my notes expressed considerable doubt about the why of it. Of interest he made clear at this visit that he had renal pain that had been present for years and always left sided. My notes had never before recorded this fact. As for RTA the idea was foreign and unacceptable to my mind.
Diagnosis
 Surgery
Surgery
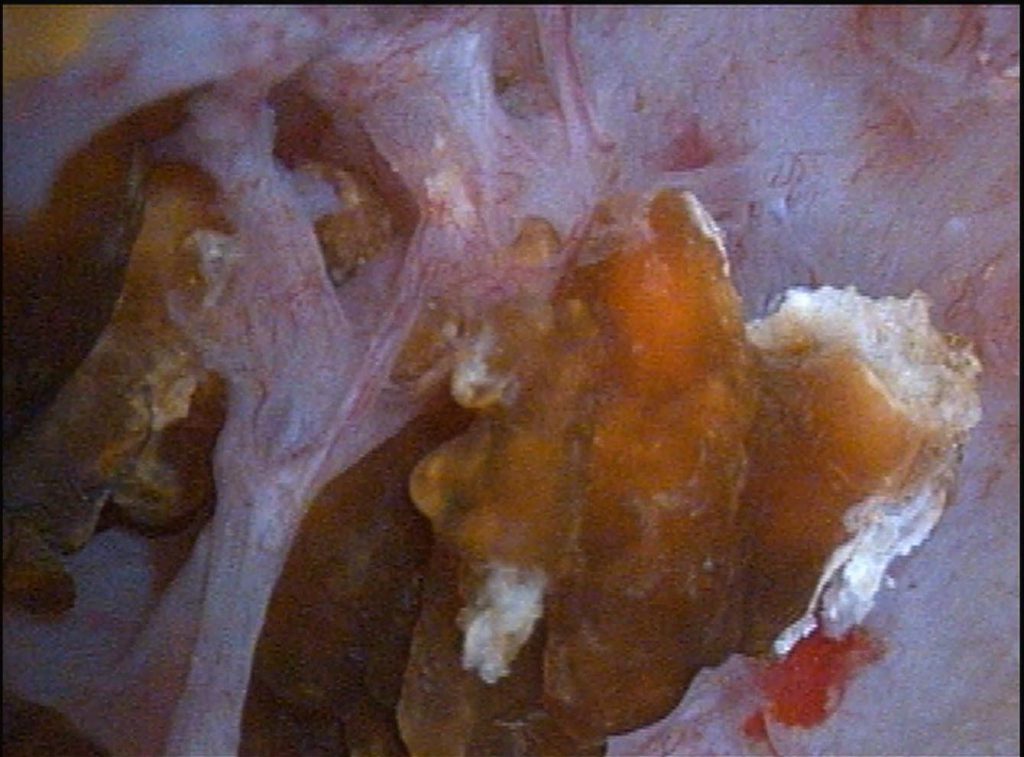
At PERC he had the characteristic papillary changes of MSK. The papillae in the upper pole of the left kidney contained many cavernous spaces filled with stones.
At the left of the panel to the left of this text a stone peers out from inside a dilated duct, and a larger one, in the middle of the panel, reaches out of its duct, one side of it a jagged glittering blue white.
The tissue itself is diaphanous and layered, seen well in the partition between the stones in the left and middle ducts in the image. Beneath the surface lies a maze of dilated ducts filled with stones; the openings to two of them are at about noon and 1 pm in the picture.
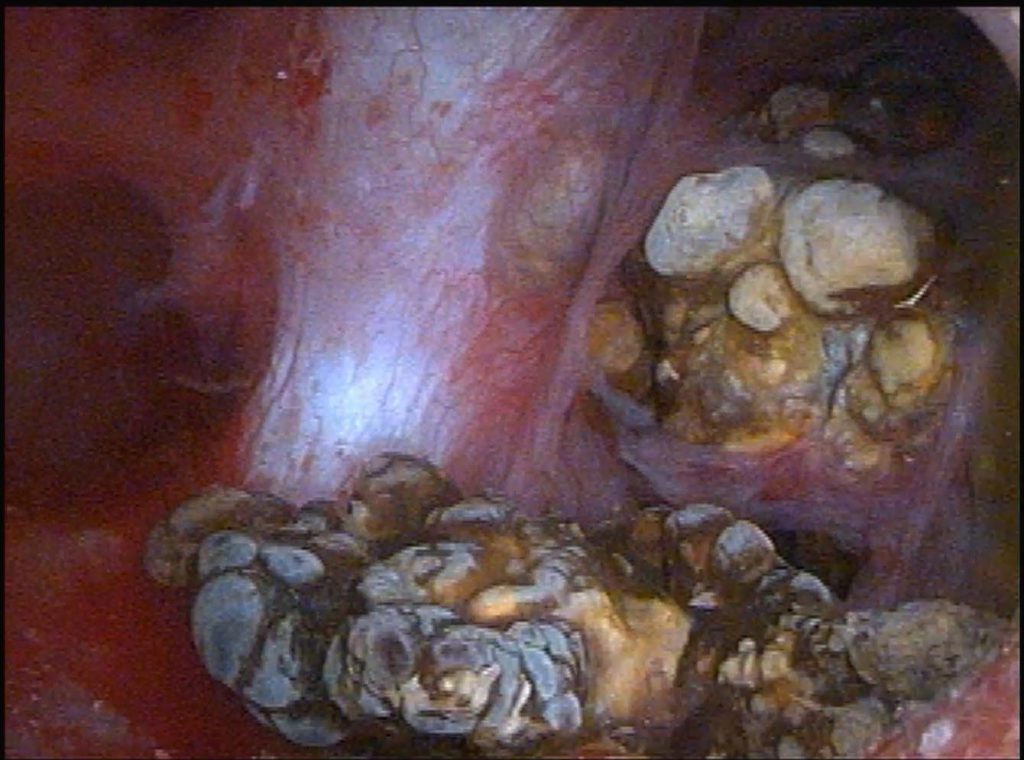
Imagine a family come to farm the land above the hollow shafts and tunnels of an unsuspected cave. As with time minerals in the sluggish waters consolidate into crystals and the crystals into myriad strange shapes unseen, tubule fluid in these dilated ducts – we believe – fills with innumerable stones that appear enormous in the magnifying lens but are to the eye unaided tiny.
Seen in another papillum, at right, is the opening of such a duct and to its immediate left, facing toward the leftmost light colored stone in the tunnel – as though it were a mirror, a bulge below the shiny covering of the papilla marks another tunnel not yet opened and filled with stones.
We have shown MSK in another article, and detailed there the laciness and tunneling, and even in a movie the unroofing of some tunnels so one can peer down into them from above. Likewise we have already spoken about how ductal dilation comes about in the development of the kidney which goes awry.
Pathology
A biopsy from the papillum stained with Yasue stain shows no crystals. The linings of the large collecting ducts – enormously larger than normal – are not in a single layer of cells but three layers or more. These are two of the three diagnostic traits of MSK. The third 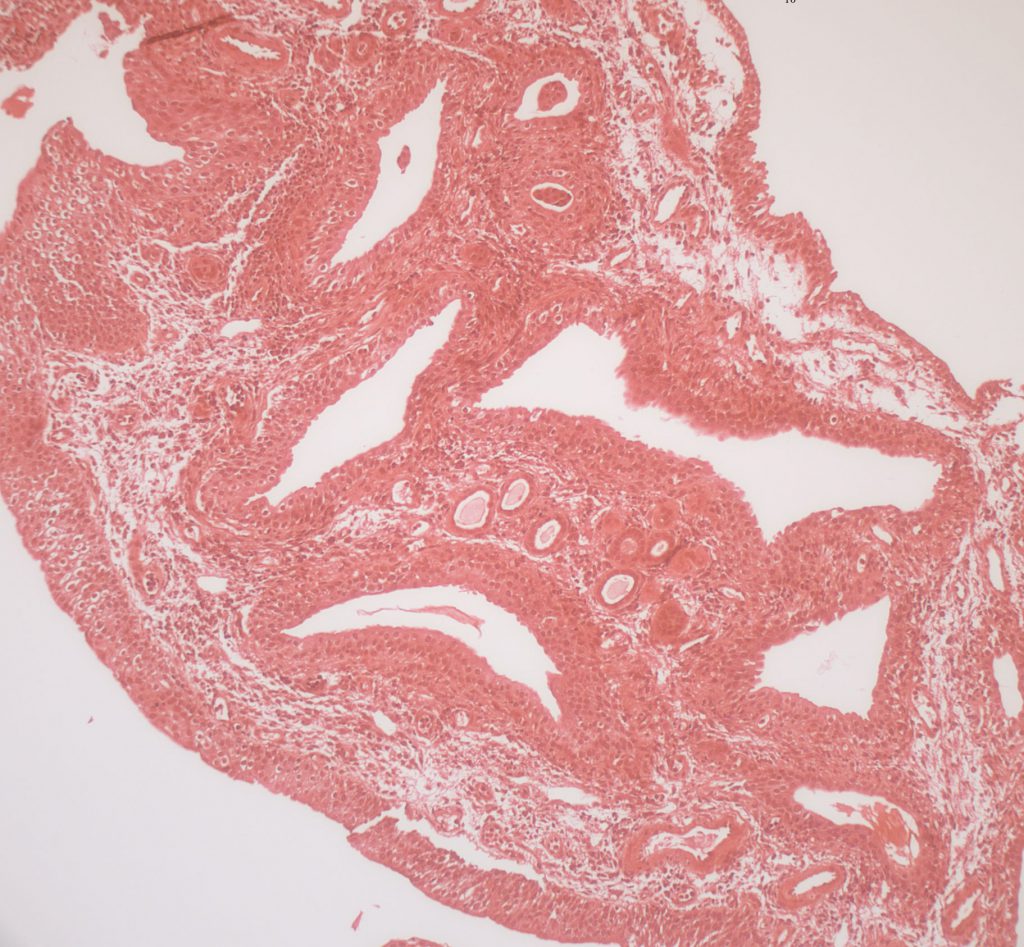 diagnostic trait is the very cellular interstitium – this is easy to see at nine o’clock where the cells become lacy looking because they have the traits of embryonic cells with many extensions off the cell bodies.
diagnostic trait is the very cellular interstitium – this is easy to see at nine o’clock where the cells become lacy looking because they have the traits of embryonic cells with many extensions off the cell bodies.
Some idea of the tremendous enlargement of papillary collecting ducts can be obtained by looking at the four or five smaller round ducts down toward 6 o’clock and ringed about the upper surface of one of the huge ducts. These are also papillary collecting ducts and even though they are much smaller than the huge ones they are still somewhat larger than normal. Even these are lined by multiple layers of cells – this is hard to see at the magnification of the slide.
So, given the enlargement of the smaller round tubules, and that the dilated ducts are perhaps 10 times larger, the overall dilation of the large ducts is perhaps 15 to 20 times normal, an immense distortion.
Fifth Visit
Looking Back
After the surgery, and with the truth of things made evident, I thought about the years behind and that I had not taken very seriously the idea of MSK first raised by an IVP long ago. In his recent CT he had one sided massive radiographic nephrocalcinosis. This itself could have been the reflection of any number of conditions – calcium phosphate stones only on one side, for example. But his stones passed were mainly calcium oxalate, a fact more evident in his recent than his first visit. Medullary sponge kidneys more often have calcium oxalate than calcium phosphate stones.
There was also the slightness of his urine abnormalities compared to the extreme amount of renal mineral deposit. If indeed stones in MSK form as in a cave, from stagnation of tubule fluid in blind end dilated ducts, then even the most normal urine chemistries are sufficient because calcium oxalate supersaturation is near universal whereas calcium phosphate supersaturation is not. I could have been, perhaps, more prescient.
Treatment
Given that no trials have ever focused on this uncommon disease, and the formation of stones may well be simply spontaneous crystal formation from supersaturated tubule fluid stranded and stationary in dilated ducts, my tendencies would have been simply to lower supersaturation by any means possible. This would lead me to a 1,200 mg calcium, 1,500 – 2,300 mg sodium diet. Compared to present calcium and sodium intakes, the higher diet calcium will lower urine oxalate and the lower sodium intake will prevent a rise in urine calcium. Likewise the higher the fluid intake and urine flow the lower the supersaturation. And, given that refined sugars raise urine calcium abruptly in even normal people, I would prefer he avoid them. In other words, I would have wanted the kidney stone diet which is the modern healthy people diet for Americans. Finally I would have eschewed oxalate loading from any source.
I had at the beginning used potassium citrate for his treatment, based on a pattern of thought I would no longer be guided by. Yet he remembered it and that his stones seemed less when he used it. So at his request I added it back. He is due for a new low radiation dose CT in a while to see if his kidneys are really stone free, and I suggested he wait to use the drug until then.
This Case in Large Perspective
Our Prior Work
As against our other reported cases, this one would be as abnormal as any we have seen. The duct dilation, multilayered epithelium, and lacy aberrant interstitial cells are remarkable. Our surgeon had no difficulty in diagnosis during PERC because the dilated ducts and abnormal papillary shape were obvious.
Yet, an experienced clinician was surprised altogether, even being one of the authors of our paper.
You who practice, beware: MSK is as easily underdiagnosed as overdiagnosed, renal tubular acidosis likewise, and the lesson is clear. Radiographs are of minimal use. Only ureteroscopy can be definitive, as can PERC.
One takeaway is the combination of calcium oxalate stones and minimal urine stone risk abnormalities with massive nephrocalcinosis.
Because stones seem to form in stagnant chambers the lower the supersaturation the better. Likewise for higher urine citrate – as an inhibitor.
New Published Work
We have already commented in our paper about the probable cause of MSK as a developmental defect of the kidneys and urinary tract. But recent work since our paper suggests MSK can be associated with other genetic diseases. This new paper reviews prior reports of linked genetic defects with MSK and adds to the list. Of 143 cases of MSK 6 – all men – had associated defects that involved the heart of brain or – on one case – was Marfan syndrome. In two cases relatives also had MSK. A prior paper from this group found an additional 7 patients with developmental disorders. This makes a total of 13 of 143 cases or nearly 10%.
My personal concern with this work is how MSK was diagnosed. In the paper, and confirmed by private email correspondence with one of the principal investigators, it is clear that contrast radiographs were always used, so one can believe that dilated ducts were directly visualized. One hopes that as URS and PERC are needed for stone management the diagnosis will be confirmed more directly and those comparisons published. This is a very important cohort and the findings of linked genetic and developmental defects of long term interest.

I have been diagnosed with MSK, RTA, and nephrocalciphonisos. I am a 33 year old female. I have chronic renal colic and it’s over taking my life. I follow a calcium oxalate diet and drink a lot of water but the pain is always present. I hate taking pain meds as I have three children and have a husband to care for. I have 1-2 surgeries a year to clean me out. What else can I do to make my quality of life better. I want to work. I hate not working but with the pain and low energy it’s impossible.
Hi Jenelle, I would say the treatment you are getting is not enough. Take a look at this article on treatment and see where you fit in. Likewise, here is a broader look to be sure the foundations for prevention have been laid. Let me know, Regards, Fred
Very interesting Fredric, I see you are reconsidering an old case, this is a good sign, it means there’s still hope for our MSK members, all diagnosed with MSK, most make ca-ox stones, but many CaP (my sister Laura) most suffer from chronic renal colic, many of chronic uti’s/pyelonephritis, most diagnosed with nephrocalcinosis, recurrent stones, RTA, hypocitraturia, high urine Ph, hyperoxaluria but not only, many other coexisting disorders like autoimmune EDS, lupus, RA MS Raynaud’s, Sjorgren’s, Diabetes, IBS, Crohn’s, not to mention hematuria, RLS, edema and emotional disorders, depression, confusion, anxiety, etc etc, the list is much longer, our MSK is very complex.
Hi Celia, I did not so much reconsider him so much as his surgical findings reconsidered me. I am glad you have appended this list of associations to the case report; the article reviews new genetic associations from Professor Gambaro’s group as well. Warm regards, Fred
I was diagnosed with MSK in 1978 by an IVP at age 24. My life has been filled with a tremendous amount of stones and suffering and am fortunate to be under the care of a terrific urologist in Indianapolis, whom you know. Thank you for your dedication towards kidney stones and particularly MSK. I was very interested in your reference to genetics, as my daughter was born with Gitelman Syndrome, and I have always thought it was strange with both of us having such bizarre abnormalities of our kidneys. Have you come across this genetic kidney defect before in your studies of MSK?
Thanks again.
Hi Cecile, No, I have not encountered this linkage but it is now on the site with the case so everyone can make the connection. As for your terrific urologist, he is. Regards, Fred Coe
Dr. Coe, I, too, was properly diagnosed with MSK and nephrocalcinosis after my urologist did a lithothripsy to remove a 7x14mm stone, blocking my ureter. She noted that my kidney was full of Randall’s plaques and embedded stones …
As with this patient, I have severe flank pain, even thought I am not able to pass my very large stones. What is your takeaway as to stones that remain lodged in the kidneys; is it possible to the kidneys to feel pain? I have a very rough time with flank pain (I have bilateral MSK) when I do even the simplest chores around my home or grocery shopping, or even walking around for too long. Unfortunately, many medical professions believe that flank pain is ‘muscular’ unless there is an active infection or blockage.
I greatly appreciate you posting this article, as well as your expertise and continued research on MSK.
Hi Heather, Embedded stones and plaque are not so characteristic of MSK as they are of brushite stones. Be sure about what was found in you. MSK creates large cavitary cysts filled with tiny stones, and these can be unroofed. Plugged tubules can, too, but with more hazard and restraint. Plaque is not part of MSK and is rather modest in such patients. Stones are calcium oxalate in MSK for the most part. As for pain, small stones do seem to cause it, but surgical treatment has not been proven effective thus far. Pain that worsens appreciably with movement or exertion may well be muscular, because renal pain tends to be interior and not much influenced by motion – as a rule. Regards, Fred Coe
Does exercise help prevent stones? Is walking a helpful way to prevent stones? Thank you for your time.
Hi Michelle, Walking is very valuable for health but alas I know of no benefit for stone prevention. Regards, Fred Coe
Hi Fredric, I notice Heather commented above, Heather is one of our group members who has been suffering from chronic MSK pain for years, like so many others, including my sister Laura. I’ve been told that pain becomes chronic when it lasts or continues for more than 3 months. Heather, my sister and so many other group members have been suffering from this same chronic pain for years!
We’ve all been wondering for so long, what causes this chronic pain in their MSK (Nephrocalcinosis RTA stones pluggings pyelonephritis chronic uti’s). We’re not scientists, anatomists nor world renown researchers but we’re still hoping you experts can figure it out, and let us know. If not a treatment or even cure, this information would help our MSK group members afront and deal with ER Drs and nurses who continue to ignore and underestimate their pain, have the nerve to ridicule them and send them back home with no medical assistance, even in emergency cases of urinary tract obstruction, active stones and pyelonephritis with risk of sepsis! All because they’ve never heard of this MSK!
We know you’re following our group, we know you care and are trying to help with stone prevention, we trust in you for bettter MSK awareness in the medical community.
Dear Celia, Of course we do not know. Partly it may be inflammation, from stones, from surgeries, from infections. A huge question is whether surgical removal of plugs or unroofing of cysts will improve pain or not. Such a trial would be invaluable in planning for treatment. Prevention is my main theme and I see no reason why it should not be pursued with vigor for and by every patient. Many are frustrated by what appears to be inconsistent results, but I believe that much of those inconsistencies can be remedied. I am especially interested in what patients can do for themselves to bring that about. Regards, Fred
Dr. Coe, I was told by a Urologist that I have MSK in my left kidney (25 years ago). It has never been mentioned since. I do not recall ever having a completely clear CT in the past thirty years, although I may have had ONE a month after lithotripsy. The largest stone that I have had to my knowledge was 9mm x 5mm x 3mm and I passed it at home–I had four small children and there was no time for an ER visit or surgery!. Although I have had many stones, they are small. Some of them causing me A LOT of pain and eventually needing some help getting them out.Most I have passed. I am usually very active but find this current battle with stones has left me discouraged and exhausted. The 5mm stone and 6mm stone that landed me in the doctors office in early December were gone December 29. However there are three “new” tiny stones. Is it possible MSK is causing the pain? Or movement of the stones.? I am about to get a “million dollar” work up because the doctor insists these stones are not causing my pain and nausea. I am baffled and frustrated. The nausea and pain are real and thankfully not due to a neoplasm-workup so far unremarkable. My blood calcium is low right now. (anorexia due to nausea?) I was on a low calcium diet for 16 years, (late 80’s – 2002) then the doctor put me on low oxalate due to my age with the risks of osteoporosis outweighing the benefits of the diet. It didn’t seem to change the number of stones I am making. The only thing that seemed to help was HCTZ and increased water. Could MSK be the issue? Thank you for all of your wisdom. I just discovered your site yesterday and am reading as much as possible.!
Hi Victoria, Firstly I am always sceptical of MSK diagnosed by CT alone; usually all that is seen is numbers of tiny stones that could be from any number of causes. One clue is that the stones are indeed calcium oxalate and that the calcifications involve more than the tips of the papillae – subtle enough distinction. Whatever about MSK you are forming stones and have pain and it is very hard to say stones are not the cause of pain when present. Usually prevention of stones will gradually obliterate the pain. Take a look at these two approaches to prevention, and be sure they have been done right and abnormalities treated. From your note I am not sure. Putting it altogether Stepwise approach to stone evaluation and treatment Regards, Fred Coe
I had 5 golf ball stones removed in April, 2016 from my left kidney. Stent placed for 30 some days. I had emergency surgery a week later because my stent became blocked. In Sept. 2016 I had ESWL with no stent. I passed a few stones. I started in August on HCTZ (fluid pill). X-Ray in late October showed 1 small stone in each kidney. October thru December 2016 I passed 19 large stones on my own (10 to 13mm). 8 of the stones were tested in November came back as being 80% Calcium Phosphate. I have changed my diet, low sodium, low protein for the last 3 months. Should I be taking another supplement along with the fluid pill and do these kinds of stones for quickly?
Hi Jody, I doubt you have MSK even though you reference your comment to that article. Given such vast amounts of CaP stones (I presume the 20% is calcium oxalate and that brushite is not named) I would do low sodium and high fluids and a thiazide type diuretic – this last does nothing to urine volume, it lowers urine calcium excretion by a direct effect on the kidneys and only works well when salt intake is low. I favor chlorthalidone 12.5 mg daily and check if urine calcium is low; if not 25 mg daily. Low protein means normal, 0.8 to 1 gm/kg/day, there is no purpose to restricting below the normal requirements. Calcium intake is important for bones, and when high and with meals reduced urine oxalate. Take a look at the right article for you. Regards, Fred Coe
Hello,
This was a fascinating article! I had an 8mm stone removed from my left ureter by ureteroscopy on Jan 11th, after having occasional agonizing pain episodes for 2 years. I found out in October that it was a stone and tried to pass it but failed. I’ve still be getting pain in my kidney and the same kind of ‘twinges’ in the left side of my abdomen that I had when the stone was still there. Is that normal for a while?
Thanks!
Hi Deana, The pain may be from ureteric irritation and should abate. If it does not let your surgeon know who will figure out the problem. Regards, Fred Coe
Hi Dr. Coe,
I have been diagnosed with MSK (about 5 years now.) Medullary Nephrocalcinosis in both kidneys, renal pyramid calcification and scarring in the upper and lower poles of each kidney. I currently have 3 kidney stones which are named, though they are small at the moment. I have gotten a UTI once a month for the past 4 month. My Issue is pain. I don’t like taking pain medications either. I have a life to live. but I find if I don’t take something then I don’t sleep at night. I have to apply pressure to my kidneys in the form of lying on a pillow, and placing a heating pad on top of it (the heating pad really help). I have read MSK is supposed to be a “benign” disease. but I am constantly in pain. all day every day. It waxes and wanes but is always there and drs look at me like i’m some kind of drug seeker. Even though I take my potassium citrate as ordered and only take 1 pain pill at bedtime and that’s it. ibuprofen during the day. when I run out of medication, its unbearable. My questions is A.) Is this a finding that you periodically see in the setting of MSK B.) How do I explain to my Dr. what I am feeling in order to try to get some help. This is no quality of life. I recently moved. So trying to find a new Dr who understands hasn’t helped either. Thank you so much for any advice.
Hi Candice, You are part of a huge ‘small stone pain’ community that raises the issue of whether surgical removal would be a benefit. In your case I have some doubts about what is wrong. The polar scaring sounds more like reflux nephropathy than MSK and possibly you have infection. Your physicians may want to pursue the latter issue as infection can lead to pain. Your UTI history supports my suspicion of infection in the kidneys. Perhaps the stones are infected. The key physician you need is a very skilled stone oriented urological surgeon. Regards and hopes for getting the right person, Fred Coe
Hi Dr. Coe, I was recently diagnosed with MSK, and I am relieved to find your website. Thank you so much for it. I did not think to question my diagnosis but after reading your site, I want to make sure that the diagnosis is accurate. I had a CT scan w/wo contrast and the findings are as follows: “left kidney there is a punctuate calculus in the upper pole and a punctuate calculus in the lower pole. Right kidney there are multiple small and punctuate calculi in the mid and lower kidney. Largest inferiorly measuring up to 3mm. Calculi are seen mostly at the medullary pyramid tips. Urographic images demonstrate bilateral multifocal renal tubular ectasia. There is no hydronephrosis. There is no evidence of renal mass. The urinary bladder shows no focal finding. Impression: bilateral MSK with bilateral nephrocalcinosis.” My urologist also performed a cystoscopy which was negative. Should I have a second opinion or further testing to confirm the MSK diagnosis? Also, separately, the CT scan revealed a pericardial effusion, which led to an echocardiogram. The echo found my mitral and tricuspid vales to have mild regurgitation and trace regurgitation for the pulmonary valve. I was shocked and overwhelmed by all these findings at once and the link on your site to the NCBI PubMed gave me a much needed possible explanation. I’m 50 and have always led a very healthy lifestyle. Thank you.
Hi Diane, I gather you had a CT with contrast and dilated terminal ducts could be seen. That is sufficient for diagnosis. Prevention is much the same as for all stone diseases and you should pursue it. I hope things go well with your cardiac issues. Regards, Fred Coe
Thank you so much for your answer. Yes the CT scan was with contrast. I don’t recall having a 24 hour urine test as is recommended on this site (however the doctor did send a urine sample to a lab). Is that something I should request from my doctor? My doctor told me to drink at least 2 1/2 liters of water each day, preferably with lemon, follow a low oxalate diet (I am eliminating the big offenders), and come back in one year. Thank you again and kind regards.
Hi Diane, As much as I admire the diagnosis from an infusion CT I do not much believe in treatment without 24 hour testing – too random and often ineffective. The high fluids are always a good thing. The lemon is simply for taste as if one really needs the potassium and citrate lemons contain one would need a large amount of them and there are better ways to do this. Without a 24 hour urine how could you know if citrate matters at all. Regards, Fred Coe
I was diagnosed with MSK about a year ago however I’ve been dealing with kidney stones for about 8 yrs now. I’ve had several surgeries to remove stones that were too big to pass, my scans of my kidneys look like snow globes.
The information I have found on this website has been invaluable. I have been trying for years to find someone who will just listen to me. I live in Nashville, TN and have had absolutely no luck finding a doctor that will just listen and help me come up with a plan to help manage everything that accompanies my MSK diagnosis. Do you ever recommend doctors to patients? The best advice I have received is that I will most likely have to go out of state to find someone who can address my issues.
Thank you for creating this website!
All my best,
Carolyn
Hi Carolyn, Vanderbilt is in Nashville, and Ray Harris is a verys smart nephrology professor who I admire. I do not believe he personally does much kidney stone work but he will know the right person. Let me know, Regards, Fred Coe
Hi Dr. Both my kidney’s are filled like case 4.I have had chronic flank pain and passing stones almost daily since 2006 .I started passing large up to 10 mm stones since 2/20016 I passed 22 stones in one day last may .My CT said innumerable non obstruction calculi through out my collecting systems forming a cast beyond the medullary pyramid. My left is worst but my pain is always right flank .One thing I believe causing increase in passing is medipose .I have passed over 100 stones since 2/2016 all some up to 10 mm.My Dr Euro whom I love wanted to do surgery right away to remove a 9.1 mm in length in my mid left collecting system that was just one Example my CT said. I have send your case study to My Uro. I have no life passing stones 24/7.I am very afraid of surgery again I had septic Shock after not my Dr but same hospital Dr stuck stents in with a known infection in 2012 I am only here by the grace of God.I have been infection free since 2013 now but every surgery I end up in hospital with infection my body also rejects stents.I would like you to email me so I can send you some pictures of my large very weird looking stones and my Ct you can read that.I am a good case study for you. My daughter has MSK had 3 stones at 4 years old I believe she got them in my whom because she dose not make them as I do but her infections scare me because of sepsis. I shared your study with other Msk people whom all have chronic pain like me . Thank you Lori
Hi Lori, I suspect you do have MSK and you have obvious serious infection and pain problems. I do not understand why there is not more effort at prevention and perhaps some surgical management of the disease. Ideally you would get your care at a university medical center. Where do you live? What were your stones made of. Given the sizes of your stones, I am not sure the whole problem is MSK. Have you had 24 hour urine testing? Let me know. Regards, Fred Coe
My MSK was diagnosed about 20 years ago after IVUG. Now my doc just does an x-ray once a year to evaluate if I am making any stones two large to pass. He did order a 24 hour urine at my request.
Hi Dr. Coe,
I was diagnosed about 8 years ago with MSK and have had lots of trouble with doctors feeling the need to do anything about it. After years of being told nothing needed done I insisted on seeing a specialist. I was referred to a urologist. The urologist decided nothing needed done since all my labs came back pretty much normal. Yet pain and stones and more growth can be seen in the many CT’s that have been performed over the years. How can a patient push for better care or further testing to confirm MSK is the true case for further preventive measures. It really concerns me as my 11 year old has presented similar problems and similar lab work. I do not wish for him to go through the same lack of care I have received with no avail to a true diagnosis or care plan.
Hi Pamela, Of course the problem is twofold. Perhaps your physicians are correct in their assessments – about surgery especially. Perhaps your condition needs consultation beyond what you have already obtained. However given more stones, the latter may be the more correct – from so far away I have very little to go on! The obvious answer is consultation at a kidney stone center. I do not know where you live, but if you want to tell me I can make suggestions. Regards, Fred Coe
Hello Dr. Coe,
I just read your MSK and the new info that MSK may be associated with other genetic disorders with the study done on men. I have MSK,diagnosed by 2 different Drs after 3 ureteroscopy. I also was born with an incomplete pericardial jacket which caused a large pericardial cyst requiring thorocotomy and lobectomy.it was explained to me this was an embryonic defect. I am female. This article hit home! I am working with my nephrologist on stone prevention,and following advice from both you and Jill Harris. Knowing that I have an associated issue of the heart in connection with MSK, must I work harder at prevention?
Hi Elan, Indeed it would be a benefit to avoid any need for surgery in the future, so prevention is especially important. Regards, Fred Coe
Hi,
my stepson has had MSK since he was a teen and is now 29.. Over the years things have gotten worse some days he can’t get out of bed because of the pain. His urologist doe not have a plan other than remove the stones when they are too big to pass. He also won’t refer him to a nephrologist. We desperately would like to find someone who could help. We live in Hickory, NC. Any suggestions would be great.
Hi Joshua, In North Carolina you have Duke University, and they have a brilliant kidney stone program. I would take him there. The best stone surgeon there is Dr Glenn Preminger. He is outstanding. Regards, Fred Coe
I live near Knoxville, TN. My stones are deeply embedded in the meat of the kidney. My doctor has tried various surgeries to remove them. He suggested to leave them and focus on a diet plan. I am a 43 year old female. Should embedded stones be left alone to grow? I am full of stones on both sides.
Hi Mandy, I agree with your physician – focus on prevention. But prevention is a whole process, so please follow a plan. Here is one of my favorite articles. Regards, Fred Coe
Hi Fred, I am in the exact same boat as Mandy, 50 yr/old with Ulcerative Colitis diagnosed when I was 20, had lithotripsy in my 20’s never had any more issues with stones until last month, but was not followed by a Urologist as I went away to school etc. CT shows full of stones on both sides some deeply imbedded. MSK diagnosed by radiologist from CT scan and then Urologist. Awaiting 24hr urine testing, advised to follow low oxalate diet in the mean time. Seeing Urologist in 4 mos for follow up. Urologist may recommend a colleague in Ottawa look at scans to see if surgery is an option as he feels they may not be able to get to them easily.
My brother has CalOx stones for many years as well and is having success with Potassium Citrate. Do you find it strange that I would be diagnosed with MSK and he would not be? Seems weird to me. I also do not want surgery if it isn’t needed, although I want to follow the protocol in order to not grow any more. Should I have my stones re-tested? I have one that I passed and Urologist never asked to see it. Also, can you share with me the link to that favorite article you had said you shared above to Mandy as I don’t see it. Thank you!
Hi Jocey, I doubt MSK is your main disease, but if there are myriads of stones and stones are calcium oxalate perhaps you do have it. The 24 hour urine testing is not about diet oxalate, it should be about all of the factors that can affect stones: calcium, oxalate, citrate, pH, volume, supersaturations. Oxalate is just one of many remedial problems. Yes, analyse all stones. Yes, it is peculiar about you and your brother, and I suspect you do not have MSK. Surgery is done because of pain, bleeding, obstruction, or infection. Otherwise it is elective. Take a look. As I answer questions, I no longer see older ones, and cannot find Mandy right now. She is no longer above, but somewhere below. But if you can find her, the link will be in her answer and you can copy it. Regards, Fred Coe
Hi Dr.
I live in michigan between ann arbor but also close to toledo OH.
Could you suggest a good Dr that specializes in MSK in this area
Ive had MSK for 14 yrs
Just turned 40 y/ o Female.
Hi Andrea, Dr Dauw works in the stone program at Ann Arbor and I knew him in Chicago. Being close, he would be a good start for you. Let me know. Please feel free to use my name by way of introduction. Regards, Fred Coe
Hi, I live in Houston. Can you recommend a Dr. who specializes in MSK in the Houston area?
Hi Louisa, There is an excellent kidney stone group at UT SW in Dallas, and Dr Peggy Pearle has vast experience with stone diseases including MSK. Regards, Fred Coe
Hi Fred. Thank you for the excellent article. I was diagnosed with MSK in 2014 and was just told to increase fluids. Since that time, I have been pain-free, but have felt increasingly “acidic” which has caused dry, cracked, bleeding hands and feet, only relieved by drinking sodium bicarbonate upon arising each morning. I live in northern NJ and I’m having trouble finding a nephrologist with good knowledge of MSK. Can you recommend a physician in the northern NJ-NY metro area? Thank you.
Hi Jennifer, Dr David Goldfarb at NYU is outstanding and I would recommend him. Regards, Fred
Many thanks, Fred!
Hello. I too was just diagnosed with MSK in northern NJ. I have been having stones for the passed one year. I have had 2 ESWL and 4 ureteroscopies but have never passed a stone! I was hospitalized for sepsis twice. I am just trying educate myself and trying to prevent stones from forming as I get some work up done.
Hi Barbara, Please also read this article on MSK, and be sure it is the right diagnosis for you. Regards, Fred Coe
Fred,
Thankful for your information and grateful for your time xo
I have msk kidneys and am as I type passing a 3 mm stone from my left and waiting for , another surgery, on the right 6.3 mm (first surgery did not work ..)… having it removed from the back…both kidneys are active with stone movement…
If I could say one thing to any fellow friends it would be … try getting a license for weed and smoke every 4 hours … when needed … it’s not for everyone but nor is morphine.
The pain with this condition is real , with or without active stones… it is my personal goal to change the paragraph written in the text books here in Canada for future students…
I am in search of a specialist in London Ontario Canada that you might know or heard of that I can work with…who is msk kidney knowledgeable xo
I am reaching as far out as I possibly can ,I thank you from my heart
JUPITER RANGER
Hi Jupiter, A very large number of patients who have considerable numbers of stones in their kidneys have pain without stone passage, as you do. You need not have MSK for this, as a majority do not. If your surgeons have seen inside your kidneys via ureteroscopy or percutaneous nephrolithotomy and have seen the sponges, then you have it. Otherwise, radiographic diagnosis can be quite wrong. This does not matter to your main issue which is pain even without stone passage. This latter – or last – can be from passage of crystals. You can tell if it feels like stone passage but there is no stone. Sometimes the crystals can be seen in the urine under a microscope. Sometimes the urine appears milky. In others, there are no crystals and the pain is of unknown cause. Also unknown is whether surgery will help, no trials have been done. As for a referral, I am afraid I am not very worldly and do not know who might be there. Regards, Fred Coe
Jupiter…please look up Dr. John Denstedt from London Health Sciences Centre…he has been around a very long time, he did lithotripsy on me 30 yrs ago and still follows my brother for his stones.
Hi Dr. Coe,
I am a healthy 62 year old woman who was diagnosed in my 30’s with MSK. At the time I had multiple calcium oxalate stones, 75%, that were removed using lithotripsy. My urologist at the time prescribed Miacalcin or Calcitonin Salmon nasal spray which I have taken since. A recent study has indicated that these meds may have a slightly higher cancer rate
(2.9-4.1%). At this point I wonder if I really do have MSK and if I need this medication (your opinion). I realize that I should be taking calcium along with my Vit. D. My stone formation has been kept in check for the most part. Occasionally I feel kidney stone pain and increase water consumption and even use a slant board. I do not have UTI’s which are connected to MSK. I drink about 80 oz. of water daily and follow the oxalate diet. Due to acid reflux, I am careful about consuming too much acidic foods. Over the years I have had several GP’s and none of them address MSK. A recent lower middle back pain prompted me to search for a qualified urologist in southern NH or Mass. who can test to determine if stones have been forming and where I should go from here. Any recommendations? Your site has been a great resource and I thank you!
Hi Marie, I would not personally want someone on calcitonin so long term, and I do not know that it has a role in stone prevention. The longer article on MSK makes clear that stones probably form because of stagnation, so the hormone would have little role. MSK is much over diagnosed, and you may have it or not. If a urologist wants to do anything about stones in your kidneys I would advise ureteroscopy that permits direct visualization. Given little pain, is surgery even important to you? Perhaps a new CT – if none for some years. As for local physicians, Boston is the one place I know of. Dr Gary Curhan at MGH is very skilled, and he would be a good choice. Regards, Fred Coe
Dr.Coe,
My son is diagnosed with medullary Nephrocalcinosis… His physician however so is that a possibility? Stop because he don’t know what to do about that. I also am not sure if we should be looking for why he has it, genetic disposition and where to go from here.
Hi natalie, I am not sure what the diagnosis is; if he/she means multiple calcifications it may be a form of kidney stone disease. Common causes are indeed MSK or calcium phosphate deposits. If your physician does not know what to do you need a referral to a center to help with the problem. If you write back where you live I can try to help identify a possible source. Regards, Fred Coe
Dear Dr. Coe,
I apologize for the haphazard email I had a faulty keyboard. My son, 20, has Medullary Nephrocalcinosis. He has substantial pain with it. So much so when he gets a bad flare up he throws up uncontrollably and we usually have to go to the ER for meds. This last visit he had high Liver levels as well and creatine kinase was 1008! We live in Atlanta and I really need some help with this. Any information is greatly welcomed.
Thank you,
Natalie Leach
Hi Natalie, I am not sure from the name what he really has, but since you are in Atlanta I would go to Emory. Dr Vikas SUkhatme is Dean there and also a nephrologist. While he himself would not provide care for you, I know him as a remarkable and brilliant physician whose office can surely direct you to the best physician for this problem at his university. Feel free to use my name as reference. Regards, Fred Coe
Thank you.
Hello: Assisting our son (35) in finding a specialist with expertise in MSK in the West Palm Beach area after a recent diagnosis. The pain from the stone was excruciating before it had to be removed and there are still some ongoing issues, with possibly new stone formation. After reading this helpful information, we realize that ensuring an accurate diagnosis by a skilled specialist will be crucial. Any recommendations would be helpful, thank you for this informative site.
Hi Kay, As lovely as Palm Beach may be, I know of no physicians there who are specialized in stone prevention. In Gainesville Fla, at the University, there is an outstanding stone center, and the flight should be not too much to undertake. Regards, Fred Coe
Diagnosed at age 24 in 1978 with MSK, I have suffered with passing and catching over 100 stones, probably same uncaught, and dozens of procedures. I am being followed by an excellent specialist, Dr. Lingeman in Indianapolis. I have been suffering intermittently for years with left sided pain and no evidence of an obstructing stone. Since April it has been constant severe pain, and ruining my life. How can I determine if the pain is due to my MSK or another reason? I am desperate for answers. Thank you for all your studies in this area and the guidance you provide. CiCi
Hi CiCi, I work with Dr Lingeman, and share patients. In fact your name is familiar, perhaps because you were in one of his studies. You need to let him know about the pain, so he can help you. He is one of the finest stone urologists in the world. Regards, Fred Coe
Hi, I am a 36 year old female who have recently been diagnosed with msk. I have been sick since the age of 17 with severe bouts of nausea, tremors and fatigue. These episodes come on suddenly and can last anywhere from 24 hours to a week at a time. During these episodes I am bedridden, unable to do much communicating or moving around. The extreme nausea, dizziness and sometimes rapid heartbeat, is almost too much for me to handle and I literally sleep the entire duration of these episodes. They have yet to find out the cause of the spontaneous episodes and the severity of them is just getting worse. The only other thing they have found besides the msk, is a genetic condition called MTHFR. I am homozygous for the c677t mutation. I have been severely affected by whatever medical ailment I have. I have not been able to work full time in almost 20 years and have seen more doctors and specialists than I can count. The msk was actually found on accident because I requested a CT scan of my adrenals to check for possible adrenal carcinomas because my symptoms are spot-on. My family doctor still seems to think the msk is not related to the severe episodes I am experiencing. I am from Evansville, Indiana and we do have a few kidney specialist, the one he would like me to see is not available until next year for an appointment. Obviously we cannot wait this long because of the severity of my symptoms, so I am settling for someone else who can get me in sooner. I am desperate and would like to think that msk has been the culprit this whole time, because quite frankly I am tired of searching and getting no answers. Can I ask a professional opinion? Can these severe symptoms be caused by msk? Are there any diseases or medical conditions that can be directly related to msk that could be causing these symptoms? Any thoughts or opinions would be greatly appreciated! Thanks for taking the time to read my questions.
Sincerely, Jessica
Hi Jessica, I note where this mutation when homozygous is related to cerebrovascular disease and wonder if your physicians have considered the matter. Here is a reference from PubMed. MSK, if that is what you have, will not cause this. MSK is widely overdiagnosed, and most patients have calcium phosphate stones with multiple tubule plugs. I would have thought the physicians you need are expert in this mutation and in its cerebral vascular effects, but that is mere speculation as I do not know your medical situation. Regards, Fred Coe
I also forgot to add on my first entry, that I am unaware of any passed stones. I have never actually seen or filtered a stone from my urine. Also both of my kidneys are affected and do have visible calculi within calyces or pyramids, spread throughout many locations/lower poles of both the kidneys.
Hi Dr. Coe,
Oh my gosh where does one start! First thank you so much for your in depth article as it has helped me a lot in regards to my son. My son was born with multiple medical problems and has been sick since the day he was born. He has complained of stomach pain, side and back pain ever since he was old enough to talk. He was had many bouts of hematuria throughout his life. He started having kidney stones at the age of 4 or 5. In 2005 when he was 12 he had several large stones and was hospitalized with them. He had gross amounts of blood in his urine, chronically complained of side/sides, abdominal and back pain…. In October of 2005 he was diagnosed with MSK bilaterally. Since that time MSK has been questioned as to if he really has it or not. In 2009 he had an IVP at Children’s Hospital in Seattle, WA. They told us that he did not have MSK. I questioned them at the time as by this time it had come back many times on radiology reports that it was either suggestive of MSK or due to his history of MSK and that is what he was diagnosed with. A few months after he had the IVP he was back in the ER again. I spoke with the ER Dr. as by this time they know us ): … I explained to the Dr. everything that had been going on with my son and talked with him about the IVP and the results of it. He went back and looked at the many radiology reports that they had and told me that it was in his opinion from he himself looking at and reading the reports and from all the times that my son had been to the ER and treated for the same symptoms repeatedly that he does have MSK…. Now this September that diagnosis comes into question again as I was looking for a new team of Dr.’s for my son and thought that the University of Washington would be a good place to start. He had an appointment on the 16th of this month with a possible new urologist. Very disappointed in that appointment as it wasn’t even 5 minutes long, he pulled up some recent scans and told us that he doesn’t have MSK and that he has seen this happen many times. He told us that there are no calcifications along his kidneys and that the stones that he was having surgery on in 2 days weren’t big enough for that and that he himself wouldn’t do surgery as it won’t take the pain away…. So two days later my son had surgery and his current urologist removed one 9 mm one 4-5 mm a bunch of smaller ones, what she called dust (I’m guessing this might be sand) and she also told me that he had calcifications embedded in the kidney that she had to clean out…. I talked with her about the appointment with the urologist at the university and what he said. I also told her about the other radiology reports when my son was younger and the IVP at Children’s as well as what the ER Dr. said a few months after the IVP had been done. She told me that there is a broad range for MSK and everyone presents differently. My son is tired of being in agonizing pain every day of his life and I as his mom just do not know what to do for him. He doesn’t have good pain management and his PCP/Pain Dr. is a joke which is why we are looking for a new team of Dr.’s …. we have even looked into the Renal Auto-Transplant Program at the University of Utah but I can not find any long term statistics on that? My heart aches for him, I’m tired of seeing him suffer daily, I want to help him but just don’t know where to go…. What Dr.’s really know MSK and what those patients really go through who have active symptoms of MSK. What Dr.’s will not be afraid to be proactive with pain management and not tell me that it’s in his head or that he is going to be dead in 5 or 10 years as I have been told this several times or that my son is an addict…. We live in Bremerton, Washington but I am willing to take him anywhere I need to in order to get him the help that he needs (we are not rich but if there’s a will then there’s a way). His base line pain level daily at one time was a 4 and he was able to have sort of quality of life but now his daily base line for his pain is a 7-8 and he has no quality of life what so ever and he always has pain, nausea, vomiting, headaches, the same things that everyone with MSK has. He’s in his room in the basement almost all the time. He doesn’t sleep at night time (he never has not even when he was a baby). The pain gets so bad that he vomits non stop then can’t eat, the ER thinks he’s a drug seeker and won’t give him anything….. His current urologist tells us there’s nothing she can do for him if the stones aren’t blocking. I joined the closed MSK group on FB to see what I can learn from others who suffer with this disease as my son does or at least I believe he does. I see where others write about other medical problems and can relate to my son and what he has going on and has had going on. Are these problems related to MSK because it all makes sense if they are. If you could help me at all it would be greatly appreciated. Thank you so much for your time in reading this and any help that you might be able to offer, Liz Mejia P.S. my email is lizmejia487@aol.com, could you please respond to my email as I don’t know if I’ll be back on this group
Hi Elizabeth, The most brilliant stone expert near you is at UCSF – Dr Marshall Stoller. I would suggest you consult with him. The problem is very complex and he has the resources to help materially in clarifying matters and seeing a way to treatment. Regards, Fred Coe
Hi Dr. Coe. I am so happy to find a reliable site on MSK. I am 46 and was diagnosed with MSK in 2017. I need help and would like recommendation if you know of any doctor who can help me in Manila, Philippines or Singapore. Thank you for this site!
Hi Joy, I wish I could help. I lectured in the Philippines last fall and realized they have no organized lab testing for kidney stones. I do not know the situation ini Singapore. I am truly sorry I cannot be helpful about a consultant for you, but in general the major medical schools are best. Given the lack of even testing services in Philippines you may fare better in Singapore. Regards and regrets, Fred
Thank you for your reply Dr Coe. May I know where was your lecture in Philippines last fall and who invited/organized it? Maybe it can lead me to a doctor who can help me somehow here in Manila til I find one in SG. I am based in Manila. Thank you
Hi Joy, I lectured in Manila and Cebu City. The physician who hosted the events was Dr Surposa. His email was ensurposa@unilab.com.ph [ensurposa@unilab.com.ph] Regards, Fred Coe
Dr. Coe I have suffered from bilateral MSK for 27 years. The last few years are very debilitating. I have had multiple lithotripsy procedures, countless uteroscopies and a double PERC. I was going to lose my top left lobe because two stones over an inch a piece were blocking off the entire top lobe. Fortunately I have healed well enough to keep it. Last November they finally started listening to my complaints of constant pain, fatigue and nausea. I have extreme anxiety as I continue to form stones at a fast rate. I watch what I eat and have over a 3L/day output. All my levels appear good after multiple 24 hour collections.
I am having what I believe to be arthritis and bone issues. My feet and hands become stuck my hands are wrists are weak. My wrists, shoulders, knees and ankles crack constantly.
Do you have any advice for me or suggestions. I feel like I am fighting a battle without any Allies.
Thank you for all you have done sir, I appreciate you and your efforts greatly.
Hi Pauline, Is it really MSK or calcium phosphate stones with tubule plugging. You can tell from the stone type and what your physicians saw in during your URS procedures and PERC. You might ask your physicians about this as treatment and possible causes might be affected. Your stone disease does not – from this vast distance – seem like MSK. Regards, Fred Coe
Dr. Coe thank you for your response. Is IVP the correct test for diagnosing MSK and can CT scan diagnose it
I was diagnosed by IVP on two separate occasions. I have had over 65 stones of various types. These include calcium, uric acid and struvite. I have had another bilateral cystostomy and lithotripsy to remove more stones. My bilateral stents seemed to fill up with tissue making stone passing difficult. Once the stents were pulled I was able to pass a few fragments. I have flank pain daily as well as nausea and fatigue. My latest KUB shows more stones. Thankfully the largest is only 0.4 cm and then multiple others that measure less. I also have stones that
I have been in a constant battle to live a normal life. I have had so many surgeries and procedures since Dec 2018.
I am concerned that perhaps I am forming scar tissue or calcification in my kidneys.
I will ask my surgeon tomorrow about tubule plugging. Do you feel that tubule plugging would cause flank pain,nausea and fatigue therefore that may be the issue
Thank you for your time and dedication.
Hi Pauline, Your stones are uric acid, struvite and calcium – oxalate or phosphate? Uric acid and struvite are not compatible, so you must have a mixture of metabolic causes for stones – low urine pH for uric acid, and presumably calcium oxalate stones as well – and infection causing the struvite. Struvite always arises from bacteria that can hydrolyse urea to ammonia, and can be very difficult to treat as they are infected foreign bodies in the kidneys. MSK may well be present. It is not known to cause uric acid stones, but perhaps you have it and also other abnormalities. From here I cannot do much as I have no detailed information. I can say that your condition is so complex that I am sure your physicians have considered sending you to a stone center that is perhaps more limited to that disease and might offer some additional advantages. I think the nausea and fatigue may be from infection, and surely your struvite stones are entirely from infection. Regards, Fred Coe
Thank you Dr Coe for your literature! I’m 50 yrs old and in search of putting puzzle pieces together and feel like ive been doing it forever. As a child, multiple ear infections(tubes in both ears at age 8), kidney/bladder infections, pain in left abdominal(below rib cage) during running/gym class/sports. As a teen bladder and urine frequency issues, car accident in 1989 resulting in extreme whiplash, led to Fibro myalgic diagnosis. Pregnancy in 1991, abruptio placenta at 7 months and bed ridden, induced brutal long delivery at 38weeks. Second child in 1994, no issues of complications. Then began heavy flow, painful menstrual issues and had 2 dnc’s. In 1998 kidney stone removal with stent. Then 2003 accidental head trauma(while putting up kids volleyball net, sledge hammer broke and hit me full force on head) Doctor said I was fine and take it easy. Nothing was easy, full out concusion, slept for 10 days in dark room in excruciating pain, walking, talking, thinking, all altered. Months later after having nervous breakdown returning to work, CT scan said all was normal and doc gave me meds for anxiety. Also told me to change job and lifestyle, owned and operated large family fruit farming business. Menstrual problems intensified, mentally/cognitive challenged daily. Increased marital/business/family stress. In 2010-2011 left marriage/business , had hystorectomy/bladder lift resulting in repeated surgical infection repairs and clean up, think 7. Final procedure calcium kidney stone removal left kidney(passed 2 stones 2015) 2012-2016 Educated/employed as pharmacy assistant, found non narcissistic life partner while battling bankruptcy and never ending divorce issues, rejuvinating an active zest for life! Noticing seasonal flu shots left me feeling unwell, hormone therapies unbalanced, increased brain fog/memory recall lose, right side head pain, change in eye sight, felt gassed or high. Left side lump in throat said to be sinusitis now waves when I’m tired or hungry. Did Ideal Protein Weight Lose Program to reboot, lost 20lbs but underneath felt like shit. Months later H-Pylori treatment, months later 1 of 3 HepB shot for travel and Dukeral, it’s like my body did a shift to downhill fast. Four months later (Mar2018) GP diagnosed me with ME/CFS/SEID with FM. Bedridden/housebound, standing long enuf to brush my teeth was impossible. Suffering from PEM from both minimal mental and physical exersion and POTS. Put on waiting list at CCDC in Van BC set for Aug +/-2020. Then June 2018 CT scan from left flank pain complaints discovered 2.5×1.5×1.6cm staghorn calculus in right kidney w/satellite stones upper right pole, multi left renal calcifications, lower left pole/interpolation region consistant with a baseline medullary nephrocalcinosis with attached calyceal stones. Surgical Urologist required a right kidney PN procedure, left kidney ureteroscopy/laser which possibly could leave residual as she has radiologic findings consistent with MN. Surgery was Oct 15 2018 PN procedure on right kidney completed, surgeon mentioned kidney had mass infection and his focus was cleaning it up. He opted not to do left kidney to avoid contamination. Antibiotic thereafter, sick and unwell, urine cultured enterococcus faecalis after complaints of UTI in Nov. Daily dose of long term Macrobid RX’d. Stone analysis indicate struvite with calcium. In March flare of hot flashes, chills,thirsty,uti, vaginosis, abdominal inflamation, left flank pain, energy level at 20%. Follow up CT scan another same size stone in right and left stones larger. Repeat surgery Jul 15 2019 on right kidney as well as left kidney w/stents. Experienced 14 hrs of kidney spazms when urinating, stopped when re inserted catheter, removed stents and cathader 2 weeks later. Oct 1 follow up CT scan reports growth of same size stones!!! Surgeon wants to repeat surgery in early Feb and I have declined until further investigation is done, finding root cause!!! Feels like nobody is doing anything other than surgery! Internist won’t see me because its Nephology/Urology. Infectious disease Dr says remove portion of kidney where tuff to remove stone/fragment are because of sponge kidney to elimited enterococcus. GP’s hands in the air. Seems everyone agrees all my tests are normal so they don’t know what’s going on. Now Macrobid has stopped working and on Amox/clav. I went over and above heads and have an appt with a Nephologist Jan 24 and have no clue where it will go, at first he wanted to see me after surgery until I said I’m refusing because of 10 weeks later I’ll need it again. Hello! Lots of emotions/frustrations. I just want my life back, I’m tired of struggling in survival mode. My trust and faith challenged. I’m sure I’ve left holes in this story but I’d certainly help to fill in blanks if needed. Please, she’d some light on me 😇
Hi Kimber, Struvite stones form because bacteria have found their way into your urinary system that can hydrolyze urea to ammonia and CO2. The resulting alkaline pH cause formation of magnesium ammonium phosphate – struvite crystals. The crystals are infected inside and out and their growth is driven by bacterial functioning. Your surgeons are coping with management of infected foreign bodies. Antibiotics are not very helpful unless the surgeons can remove all of the infected material. Possibly things are advanced enough in difficulty your physicians may wish to consider acetohydroxamic acid – a difficult to use inhibitor of the bacterial enzyme that hydrolyzes urea. I have used it in rare cases with success but side effects are legion. All I can say from here is that perhaps they may want to consider the possibility, but knowing nothing really about the details of your conditions please consider me as a mere outsider with nothing to offer but a slim suggestion for your physicians to ponder or not. Regards, Fred Coe
Sorry Dr Coe, forgot to mention I live in Osoyoos BC Canada, on the Washington borderline. Also, I’ve noticed for years that usually every 3 months I cycle on overall health decline with alot of symptoms/flares, like it comes and goes in waves. I did a Lyme test submitted to CDC and results were negative. Lyme and CFS have same/similar symptoms. Certainly with all the antibiotics I have had, lots of symptoms have changed/lessened except for kidney issues, worse!! Also do Accupuncture, have used RX’d CBD oil 1:20 mg/ml that helps neuro. I’ve stopped all OTC supplements/vitamins as directed by Surgeon in 2018 due to the unknown . Any advice or info, I would be so greatful. Please feel free to email me at any time. Peace
Hi Kimber, This information does not help me in thinking of what might benefit you. The problem of infection stones is as I said, and available treatments, as well. Best, Fred Coe
Dr. Coe Thank you for your input. I had 2 different IVP tests done that was how I was diagnosed with MSK I have also had CT scans and ultrasounds and KUB xrays. How is it properly diagnosed I have 3 types of different stones. some stones do not show on ultrasound i have been told after passing a large stone one day after having the ultrasound.
I will ask my surgeon tomorrow about tubule plugging. Just wondering if there are stone diseases that I should be inquiring about.
Thank you for your time and dedication.
Hi Pauline, I see the duplicated note, but want to add. The plugging is not important to you right now. It is the infected stones, and as I mentioned your physicians might want to make use of a second opinion given all that has happened to date and also the variety of stone types. Regards, Fred Coe
Hello, Dr. Coe. I commented previously on your article about nephrocalcinosis. Your reply seemed to suggest I read another article of yours regarding the diagnosis of MSK, but didn’t link to one, so I searched and found this article. I’m commenting here based on the assumption that you meant to direct me here. Apologies if that assumption was incorrect. This has, however, given me some clarity. My original questions was whether or not to have a procedure to clean out all stones, or as many as possible, in an attempt to garner a few years of respite from the large, painful stones. I now recall, after having my first ureteroscopy in 2017, my urologist definitively confirming the diagnosis of bilateral MSK. My stone analyses have come back as mostly CaOx, with some CaP. A small number, perhaps 3 or 4 stones of the dozens that have been analyzed, were mostly CaP with smaller amounts of CaOx. From 2017 until late 2019, I was following a treatment plan prescribed by my urologist of Indapamide, Allopurinol, and at least 128oz of fluids – mostly water – each day, as well as discovering and learning to follow the kidney stone diet: 1500mg sodium, 1000mg calcium, 25g or less added sugar, no more than 100mg oxalate. Potassium citrate was added in early 2019. Stone growth had been little to none in those two years, but I became pregnant in late 2019 and had to stop all medications. I’m expected to deliver in late July, but will be unable to resume most medication until after I stop nursing. I have continued to closely follow the kidney stone prevention diet, signing up for Jill Harris’ course in November of 2019 and continuing with her monthly support group, as well as being active on her private Facebook page. Would a clean-out of the stones I currently have be a reasonable course of action? I repeat this question because I have reached a point with this disease that I am, admittedly, quite desperate for a few years of respite. I have three other children – a son, 15, a daughter, 12, and a daughter, 9 – whom I homeschool, and I have a full time job to boot. The stone episodes have become so frequent as to significantly interfere with daily life for myself, but I have begun noticing that it is affecting my family, as well. I suppose I am hoping that I won’t be laughed out of my urologist’s office if I suggest the possibility of having as many stones removed as possible. Please be as frank and as realistic as you deem necessary in your reply. I honestly wish I could schedule an appointment directly with you to discuss my diagnosis, best treatment options, and any other proactive measures I could take. My current urologist seems wonderful, but I’ve had four different doctors at the same practice in two years because of high turnover, so I’m never really certain how knowledgeable or truly invested in patient care is each physician (one in particular minced no words when letting me know that my concern over chronic pain and frequent stones was a nuisance). I appreciate any advice you have, or more research you can point me towards, to help me in my decision.
Hi Chrissie, The article on MSK is this one. If indeed your urologist found MSK during stone surgery then you have it. And indeed stones are usually calcium oxalate most of the time. Given the problems with urologist turnover, one possibility is for you to consult with a second opinion. I can make suggestions, but perhaps that is best done via private email. Feel free to email me Coe@uchicago.edu. Regards, Fred Coe
Dr. Coe, thank you for hosting this reply board.
I have been quietly dealing with over 120 calcium oxalate stones in the last 20 years, my first lithotripsy(18yrs ago), my physician told me that I had MSK, then retired, nothing was noted in my chart prior to his retirement. I had never heard of the disease and my replacement urologist which I have had for the last 15 years, has told me to drink more water and take my potassium citrate.
During my last visit which was a follow up after a deroofing procedure was done on both kidneys, I was told I had MSK, dRTA, and nephrocalcinosis. I have no reason not to trust my physician, but he commented that all my stones analysis showed CaP with brushite and apatite with 70% or more of the composition. My 24 hour urine PH have all been between 6.2 to 7.3.
For 15 years I was told I did not have any disease, just made stones,….alot of them. I live in the Raleigh-Durham area in North Carolina. I am willing to travel for some guidance and help with a more intensive prevention approach to my stone burden.
1. The diagnoses seem like alot after all these years…is that common?
2. Obviously I am not seeking a diagnosis on this board, but would like recommendations on a program that has a good support staff, like a dietitian involved that can be a touchstone for the diet, I did the diet it for 2 years and found it quite difficult while cooking for a family of four, and working full time. I had no books or guidance, maybe you could recommend some?
3. While I am not sure if I believe I have MSK since it seems so overdiagnosed, I certainly believe that I will spend the rest of my life with these little stone babies. I have had great success with tens units, cold/hot therapy, foam roller therapy to relieve pain, what else is out there to help with the chronic dull ache?
4. Moonstone..was a product prescribed to me at my last visit. It is an alkali powder, that is mixed with water to make a drink..If I am already above in my PH, and I am making CaP stones, should I question this?
Thank you so much for your time.
Hi Michelle, If you do or not have medullary sponges, you certainly do have brushite stone disease, a rather special kind of problem. Whether or not you also have distal renal tubular acidosis is unclear from what little you have written. But you are fortunate in that Duke has a great stone program, and Dr Preminger is a world famous expert. Just go there, and let them take care of everything. He is brilliant. Regards, Fred Coe
Dr. Coe,
I have bilateral MSk,as seen from uteroscopy. My recent CT scan showed 5 mm right and 7mm left lower pole non obstructing renal calculus, an intervals migrated ovoid shaped 1 cm calculus presental in renal pelvis( previously in calyx) demonstrating focal calyceal dilatation,and 2 adjacent 2mm distal left ureter stones without hydronephrosis( the distal stones passed a few days ago). I felt fine the next day,but now I am feeling the same burning sensation except when urinating in the groin. Does this need attention promptly? I am frustrated that the urologist’s clinical team has not returned my calls requesting an appt sooner than July,which is the Dr first avail appt. I have a nephrologist in Orlando,and was able to get an appt in July,his next avail. I passed a stone in March also,calcium oxalate. I live in the Daytona Beach/new Smyrna area,and after moving from my hometown of Tampa,I chose Drs in Orlando over the Daytona area. My questions: do the CT findings need immediate attention or can it wait until July? Do you have recommendations for MSK care in Central Florida? A prior urologist said not even the U of Fl Gainesville Drs would be able to help with my MSK.
Hi Elan, If you can use Gainesville, it is a very strong institution. The medical renal people there are numerous and talented so prevention of stones will go well. Dr Canales is a famous surgeon and scientist in stone disease there and either he or someone he identifies will surely be outstanding. Regards, Fred Coe
Elan! My daughter suffers greatly with MSK. We live in the Daytona/New Smyrna Beach area. We are desperate for info for a good urologist. If you have any info we’d be grateful. It’s awful to see her suffer so much!
Hi Dr. Coe!
I was diagnosed with MSK 10 years ago. I have had 9 surgeries with only one being somewhat effective. I only have MSK in my right kidney and it is filled with stones that are trapped in the tissue of my kidney. After the 9th procedure my urologist stated cutting the tissue of the kidney is the only way to extract the remaining stones and would be too damaging. My largest stone is 22mm with two others following closely in size. It cannot be dealt with because we risk potentially puncturing my lung to do so based on the location of it. Management of my condition was going well until two years ago. I got a kidney infection and an infected stone continued reintroducing the infection to my body since they couldn’t remove it. They put me on 4 months of antibiotics to rid the infection. Further more, they have found difficulty in identifying all types of stones but did discover some are struvite. Ever since that kidney infection I continue to deal with UTI type infections which was never a problem before. I also have a constant ache and sometimes it’s sharp. I just moved to the Detroit, Michigan area and am hoping for guidance to find a good urologist. The last time I was in the ER the doctor stated I need to understand I will eventually lose my kidney. I’m now trying to determine what is the best route. I’ve stopped the production of stones. I’m curious if the trapped stones are keeping me in a minimal infectious state.
Thank you for any and all guidance!
Hi Sarah, U Michigan (Ann Arbor) is near enough for you to use. We are further away but have outstanding stone surgery. Either way, you need expert surgical care in a stone center. Indeed stones can perpetuate infection, and certain infections can themselves create stones (struvite stones).If you wish to consider care here our number is 773 702 1475. Regards, Fred Coe