
The video is a big enhancement of this article, I recommend it.
The Structure of Kidneys
Stones form inside the kidneys and the urine collecting system. How they form matters to patients because surgeons can see formation sites during stone removal by ureteroscopy or percutaneous nephrolithotomy. The amount of such sites gives a clue as to future stone risk and also to possible damage done from crystal deposits in kidney tissue.
We cannot discuss where stones form unless you know how kidneys are constructed.
If you already know this, move on. But if you do not let’s stop here and review how kidneys are put together.
The linked article is long, so focus only on the cutaway drawing of a kidney that shows the papillae, renal pelvis and ureter. Stones grow on the papillae.
Where Kidney Stones Grow
With that in mind, and to begin with, what exactly do we know?
One thing we know for certain: clinically significant calcium oxalate kidney stones grow in human kidneys attached to ‘plaque’ – deposits of calcium phosphate embedded within kidney tissue.
Another: Calcium phosphate deposits plug the terminal ends of kidney tubules. On the open ends of such plugs – ends that face onto the urine – small rounded overgrowths form. They contain mixtures of calcium oxalate and calcium phosphate. We believe they detach and grow into significant stones – stones big enough to cause pain, obstruction, need for surgery. But we do not know for sure because we see and harvest overgrowths only a few millimeters around – too small to do much to patients.
Often we find stones attached to nothing. They bear on their surfaces no traces of a prior attachment site. We believe such stone stones form in urine – not necessarily attached to anything.
Growth on Plaque
An Example
In the left panel of this picture, the arrow touches a calcium oxalate stone growing on a 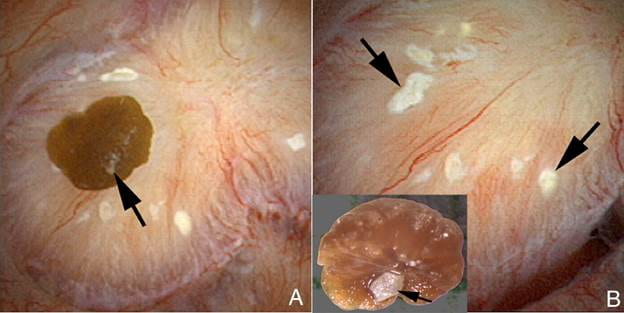 papillum of one of our patients who gave us permission to photograph during kidney stone surgery.
papillum of one of our patients who gave us permission to photograph during kidney stone surgery.
Like a shoebox filled with family snapshots the article linked to the header has many more images like this one.
The surgeon, with some effort, pulled the stone off of the papillum. Turned upside down that stone reveals a small light patch of calcium phosphate that once linked it plaque. That exact patch of plaque lies at the upper left of the right side of this picture. A big arrow points to it.
The white patch on the stone fitted exactly into the slightly darkened center of the plaque. Irregular as clouds, more plaque lies scattered about on the surface.
Evidence from Stones
Because stones that grew on plaque often take their anchoring site with them when they detach, scientists can use that site to estimate the frequency of such a mechanism. In our study detailed in the main plaque article whose link heads this section most stones found free in the kidneys bore this signature of origin. Sometimes under a microscope tubule fragments are visible.
Statistics
How often does this occur? We found growth on plaque on many of our patients but we looked for it. By contrast, others who looked merely at all comers found it less often. Though exciting controversy for insiders the final count bores me. After all, urological surgeons must eventually report on case series and get us the answer. Right now I can say stones grow this way, in people.
How Does the Stone Grow Over Plaque?
Urine Contacts the Tissue Plaque
Most of the plaque you see in the picture is inside the shiny membrane that covers the papillary surface. So long as the membrane covers it stones cannot form. But where the stone grew that membrane gave way. Urine bathed the exposed plaque and minerals from that urine formed the initial hydroxyapatite binding site that shows up at the bottom of the stone in the right hand panel of the picture.
Hydroxyapatite (HA) Grows Over Exposed Tissue Plaque
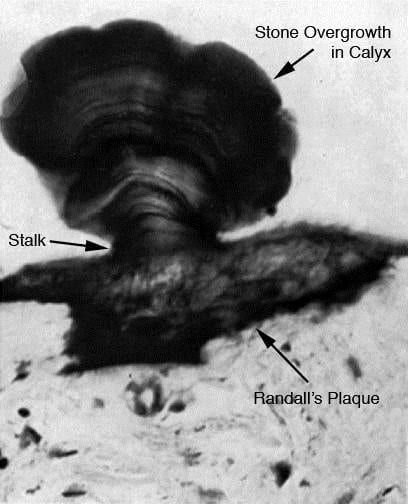
This dramatic human example of a stone growing on plaque was found years ago, and is probably the best one available.
The stone grew on a stalk that is the calcium phosphate overgrowth on plaque. This is just like the stone with plaque on its underside and its matching plaque I showed in the surgical video image.
This sample has the advantage of showing the plaque in the kidney tissue. It is the black material – calcium phosphate is stained here to bring it out.
The tissue below it is renal papilla. You can see a few end on tubules looking like round tubes.
Plaque Forms in the Loops of Henle
How could be know this? We find the plaque but we cannot watch it form.
This way. Find the smallest possible deposits, deposits so small we need powerful microscopes. Where is it? Plaque begins like all crystals, as tiny nuclei that grow, so the formation site will contain very small deposits.
That is not sufficient. Look for the site that always contains plaque even when the amounts of plaque are tiny. Possibly that could be between the interstitial cells. Or in the walls of the vasa recta, or the loops of Henle. Maybe, in the interstitial cells.
Of these, the loops won the prize. The most minute deposits lie in their walls. The blowup in the picture shows four cells lining a length of a loop segment. Between them the red arrow stands for tubule fluid. On their bottom sides run the basement membranes that face onto the interstitial space.
In those basement membranes we find plaque originates. From them or over them plaque expands between the loops and vessels until it reaches the papillary covering membrane. It lies beneath it, dormant, until some breach of that membrane permits urine to contact it whereupon a stone can form.
Plugging
How Plugs Appear

The picture at the left is a papilla imaged during stone surgery. The end of a terminal collecting duct, from which the final urine issues, is plugged by a mass of crystals labeled by the asterix. The small arrow points to a bit of plaque. The arrowheads show an eroded part of the papilla, that has been injured.
The picture at the right is another papilla photographed from the side of several plugs. You can see they are elongate, like tubes, and that is perfectly realistic. For each plug forms a crystal cast of a terminal collecting duct.
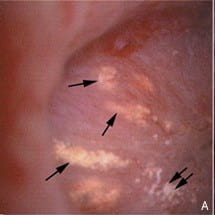
I do not show it but inside any of these plugs the lining cells are dead. The crystal plugs kill them. Around the plugged ducts is a mix of inflammatory cells, drawn to the site by cell injury.
Idiopathic Calcium Stone Formers (ICSF)
The very common calcium oxalate stone formers who have no systemic disease causing their stones tend to form their stones on plaque. As a group they do not exhibit very much tubule plugging. By contrast, those whose stones contain mostly calcium phosphate exhibit plugging and growth on plaque.
Stones From Diseases
More or less all stones arising from systemic diseases cause plugging, with or without plaque. The details will come as we consider each of these separately. This means the idiopathic calcium oxalate patients are the standouts – the ones without much or any plugging. All the rest of the stone formers plug tubule to one degree or another.
What Plugging Means
Plugs Form At the Ends of Nephrons
Crystals of the sort in the table plug the terminal ends of nephrons. Those of you not physicians probably know – or can now learn – nephrons are tubes that begin with filtration and end in a final urine. Kidneys each have about one million of them and their combined small drops of final urine make up what we produce each day.
That final urine leaves the nephron, obviously, at its nether end – furthest from its beginning at the filtration point. We name that final short end – a few millimeters long – after its first describer: the duct of Bellini (BD). It looks different from what comes before it, and in it plugs form. Just above, the inner medullary collecting ducts feed into the BD. It is in BD and IMCD we find the majority of plugs. Rarely, crystals plug higher up, in the outer medullary ducts or cortical collecting ducts, or even the loops of Henle. So plugging occurs more or less at the ends of nephrons.
Tiny Overgrowths Form Over the Ends of Plugs
Small overgrowths over the open ends of plugs may detach and grow into stones. The idea has obvious merit and reasonability. But unlike stones on plaque that often achieve clinically relevant size – 2 to 3 millimeter, plug overgrowths are often less than a millimeter. I believe these tiny knobs do become stones. We need a test of this idea and I cannot think of one. For example, might we imagine a mark like detached plaque?
They Injure and Obstruct
But this is to simplify matters too much. The many nephrons join each other as small streams and rivulets join along their ways to make larger channels that themselves merge, gradually forming, perhaps, a mighty river, even. So on the surface of each papilla one finds a few dozen BD and given nine or ten papillae in a kidney a few hundred to drain the fluid of a million nephrons.
This means what it would mean to dam up the outlets of many small streams – fluid would back up, pressures, therefore height, increase. One might expect that high up in the nephron, far from the dams, tubules would dilate or other signs of injury appear. More; unlike such an outlet living cells line BD that crystals might injure. This means we should expect signs of injury, low down and high up, and we do. Injury is easy to document.
Injury From Plugging
Low Down
The main plugging article makes clear that plugged tubules sustain a lot of injury. The lining cells are absent. Crystal plugs fill the whole interior. They adhere to the basement membrane on which the lining cells once rested and through which they once drew their nutrients from and gave up their waste products to the kidney circulation. Said another way, a tube lined by living cells becomes a mere tube filled with crystals.
The cells around the tubule, those interstitial cells I wrote about in the section on plaque, know what happened. They wall off the destroyed segment in fibrous tissue. The process of such walling off is a form of inflammation. These cells respond to the cell destruction so as to isolate the problem in scar.
All this happens in the papilla, the very end of the line from nephrons and the final urine. Surgeons can see the plugs and the scarring and deformity from inflammation. In the common idiopathic calcium phosphate stone formers plugs are numerous and small. Plugs are sparse in those who make brushite stones, but very large. Renal tubular acidosis causes small deposits but so numerous as to severely damage the papillae.
The main article shows pictures and a video of all this damage. What matters here is that plugs certainly damage papillae and lead to scarring.
High Up
As a rule, papillary injury rarely results in loss of glomerular filtration, the life sustaining function of kidneys. Even so, biopsies from the kidney cortex where the 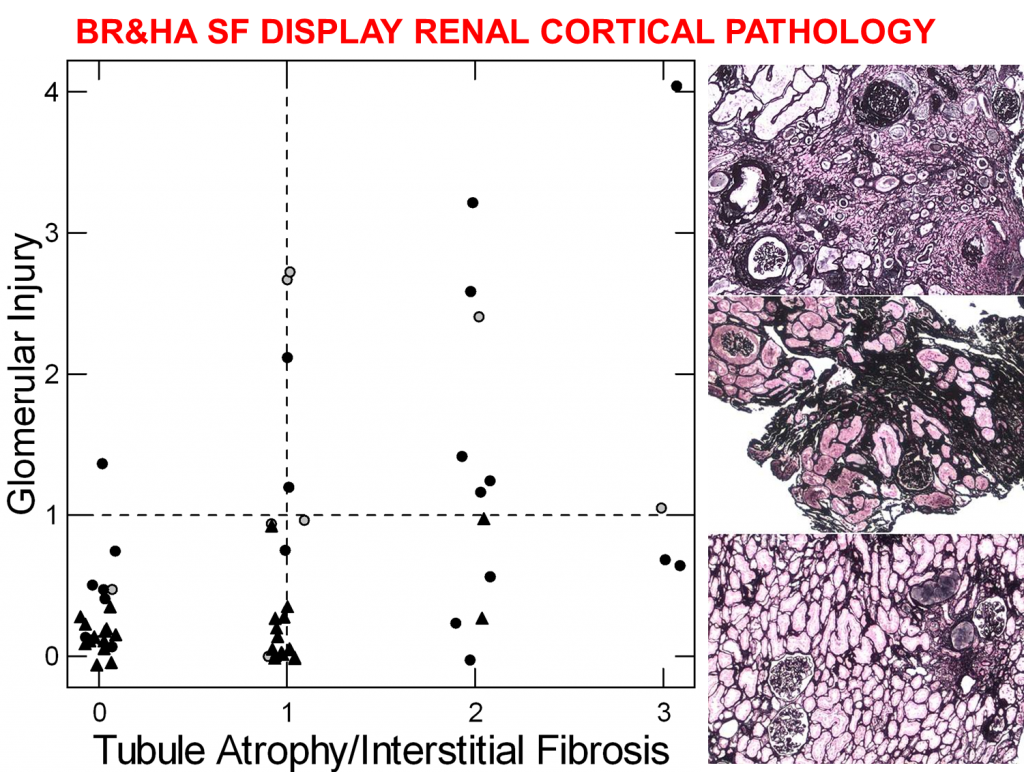 glomerulae reside show more injury in patients with plugging than in those with just plaque.
glomerulae reside show more injury in patients with plugging than in those with just plaque.
We did not put this picture into the main article about plugging. For the brushite (solid circles) and HA stone formers (open circles) in the table we obtained biopsies not only of papillae but also the kidney cortex. Likewise for the idiopathic calcium oxalate stone formers (triangles) who make only plaque.
The scores along the baseline – 0 to 3 – are grading done by a skilled kidney pathologist who know nothing about the patients. They grade tissue injury in the tubules and interstitium of the cortex. The vertical score grades injury to the glomerulae themselves.
ICSF had low scores for both, mostly 0 and 1, as one finds in normal people. A sample of their kidney is at the bottom right – even if you know nothing it looks good.
The HA and BR stone formers had almost all of the glomerular injury and tubular injury scores above 1. Their tissues, right middle and top, respectively, look more complex and irregular. Scarring and loss of nephrons are apparent to those trained in this work.
Despite scarring and injury, kidney function as we usually measure it does not differ between these three groups of patients. Neither are they lower than normal people. Perhaps kidneys have considerable reserves. Our estimates of kidney functions certainly are crude and may not detect injury until more marked than this.
In more extreme cases, however, such as with RTA, the bowel diseases, and PH1, kidney function goes down and some patients ultimately require dialysis. So when severe enough papillary injury seems very dangerous to health. What saves most stone formers from kidney injury may well be simply that without a systemic disease plugs do not damage papillae above some critical point.
Uric Acid Stone Formers
We have thus far concerned ourselves with calcium based stones but one study on patients whose stones contained uric acid belongs here. One might expect uric acid to plug tubules or perhaps crystallize in the bulk urine. In fact, plaque was about as impressive among the uric acid stone formers as the calcium stone formers, and plugging, too. Plugs could not be analysed, and might have been uric acid or calcium salts. The authors did not say where they found stones growing.
Free Solution
No link arises from the bold heading as no article considers stone formation from simple crystallization in tubule fluid or urine. Plaque, plugs, stone growth on plaque, and even overgrowths on plugs all arise on cell or fixed crystal surfaces.
But several conditions may illustrate free solution stone formation – medullary sponge kidneys and cystinuria.
Medullary Sponge Kidneys
Whereas these patients indeed pass kidney stones of usual dimensions, their sponges often fill with vast numbers of tiny round stones that do not adhere to the lining cells nor appear to evoke any inflammatory response. Their internal laminated structure of calcium oxalate crystals with organic matrix much resembles stones that form on plaque but without the telltale apatite anchor site. Their roundness, tiny size, large numbers and failure to adhere to chamber walls speak to simple crystallization in supersaturated tubule fluid dwelling overlong in stagnant blind end cysts.
Although we offer free solution crystallization as the cause of these many tiny stones, urine supersaturations themselves are meager compared to most stone forming patients. We presume any supersaturation could create these microliths given enough dwell time in cysts.
Plaque is very sparse, and BD and IMCD plugs as well. More or less they are rarely found. Routine kidney stones are usually CaOx and have the usual size range. But because of the cysts filled with tiny stones CT scans show what appears as many stones or tissue calcifications – so called nephrocalcinosis.
MSK presents us with two separate issues – the stone and the disease itself.
Because this chapter concerns how kidney stones form I choose to delay the question of MSK biology into a later time. But the stones fit well.
Cystinuria
This amino acid crystallizes easily. We find cystine plugs but they do not adhere to tubule walls and cannot anchor stones. Plaque is scant. Tubule plugs of calcium phosphate are surprisingly common. Dilated IMCD and BD also are common as is inflammation and injury around plugged ducts and high up in the cortex. In fact, cystinuria seems to damage kidneys enough that clinical measures of kidney function show reductions.
But even here, free solution seems hard to prove because the BD plugs dislodge so easily. How can be know if one or another simply lodges among papillae and grows? Likewise for tubules. We can say cystine crystallized in tubule fluid but what about the calcium phosphate plugs. How do we know if they somehow condition tubule cells to promote cystine crystallization.
Summary of Chapter Two
With perhaps one or two exceptions, stones seem to grow on some kind of anchoring surface – plaques or plugs. Exactly how plaque and plugs form, and what we might do to reduce their formation are both open research questions. SImple crystallization in free solution may produce plugs but given the biology of tubule lining cells that hypothesis would be difficult to test. Reduction of supersaturation remains our main treatment for the stones themselves. Because plugging causes cell death and tissue inflammation treatments to prevent plugging may have clinical value apart from stones themselves. The hypothesis that reduced supersaturation might reduce plugging could be tested, in principle.

Hi Fredric, thank you for this article and first 2 chapters to your book, which I’m obviously reading and following. Many of our MSK members have also been diagnosed with RTA nephrocalcinosis recurrent stones and pyelonephritis/sepsis. Many suffer from bowel disease and/or autoimmune connective tissue disorders. Research in MSK has already associated EDS to MSK but no other systemic connective tissue disorders or vasculitis. I’d be interested to see more clarification in this respect.
Dear Celia Grace, Thank you for being such a fine reader and proponent for stone formers. The book has been a trial as I am after education toward understanding as opposed to mere information, and the way is hard. I am now writing up primary hyperparathyroidism, and soon the bowel diseases and RTA. We published almost all the tissue work ever done on these so the articles are long overdue. Warmest regards, Fred
Hello Dr Coe. I had lithotripsy almost 2 years ago and just had a cat scan that revealed a 3 mm nonobstructing calculus in the upper pole of the left kidney. I was unable to catch any fragments of the stone that was dissolved via lithotripsy, so I have no idea what sort of stone I have. I do know the first stone I had dissolved was NOT evident on X-ray, which I think suggests a uric acid stone? I am keenly interested in treating this stone with potassium bicarbonate and/or potassium citrate to see if it will dissolve on its own. Where might I find guidance on what dosages to take and how to monitor my urine output? Local doctors here seem only interested in subjecting me to lithotripsy again. Any suggestions you might have would be greatly appreciated, including physicians in the central Florida area who might be willing to treat me with meds. Your thoughts? THANKS!!! Janet
Hi Janet, Try this article and its relatives. For those without a stone analysis one needs to guess. If your urine pH is not consistently low – below 5.5 uric acid stones are not likely. Above all I would pursue a complete evaluation as the articles suggest and see what comes up. RIght now I sense inadequate information. Regards, Fred Coe
Hi Dr. COE
I have imbedded calcium oxalate stones 17 on rt and 18 on left sides. Would they impair my general kidney function. Should l worry about them. Also have calcification of my pancreas. That sounds serious to me but the urologist has no comment. I am a worrier by nature. I am a retired BScN.
Janet
Hi Janet, Although people who form kidney stones are more prone to high blood pressure and kidney problems than those who do not form them, the presence of the stones does not pose an obvious danger to your kidney. Specifically, in the absence of pain, bleeding, infection or obstruction one does not need to remove them. You should have serum and 24 hour urine testing to determine their cause and take steps to prevent more and growth of what is there. Regards, Fred Coe
Fred
I graduated from Pritzker 1985. Glad to see you are still working. Trying to pass my first kidney stone now. Chapters bring back memories of pathophys. Always more complicated than one would first think.
Best,
Rick
Hi Rick, Sorry! The chapters are a pretty good guide to prevention; do it because the stones can keep coming. Glad to hear from you, but I wish it was for a less miserable reason. Warmest regards, Fred
I want to thank the author of this artical for the well thought out information he shared. I am a first time “stone former” who has received a stint and awaiting removal. I do not want to ever have another blockage! I am in hopes of causing small formations to be vibrated loose and discharged in the general flow before they gain size. Thanks again. S Durfee
Hi Samuel, Wait until the stent is out, then pursue prevention as per Chapter 1 of the same book. It is very worthwhile. Regards, Fred Coe
Hey there! I know this article is several years old however I’m hoping you’ll still able to help. First of all, thank you for sharing this information. I’m beginning to understand this whole process thanks to you, as I just had my first kidney stone removed yesterday, 5 days after my first symptom. The stone I had removed was clogging my right kidney due to its 1.6cm size. I’m still waiting on the pathology results to determine the type of stone I had (not sure if stone type matters for the questions I have).
I’ve given birth to two children naturally (my last being a year ago) and the only way I know to describe the pain from a blocked kidney is to compare it to a constant contraction only much worse. While I now understand I have a higher probability of having additional stones in the future, and preventative measures aside, i have a few questions I’m hoping you could answer for the future.
1. Is there a process to discover the formation of a future stone before it gets out of hand, like the one I had was? In other words, should I be scanned every so often to make sure another isn’t forming or are you just suppose to wait until symptoms start?
2. Speaking of symptoms, could a blocked kidney cause severe fatigue, lack of energy, weight gain, etc, in addition to pain?
3. How long does it normally take for a stone to grow to a 1.6cm size, from start to finish? Days? Weeks? Months? Years?
4. Lastly, do certain stones grow faster than others?
Thanks again for the article. Very informative!
-Becca (#KidneyStoneSurvivor -haha)
Hi Becca, Stone type always matters. Stone risk for more stones is mainly got from serum and 24 hour urine studies that determine propensity to make stones. Here is a good article on that topic. Here is another. Yearly or every other year CT scanning is now more practical using ultra low dose scanners. So big a stone would take years if calcium oxalate, months to years if brushite or hydroxyapatite, months if uric acid. You make a good point about this article – I really should take some time to update the older ones, but that time is hard to find as I work full time. Regards, Fred Coe
Just finished lithotripsy broke up 11.2 mm stone and passed fragments. Waiting on analysis. I’m 71 what age my expectations for another done and how long did it take to form this one and possible future ones. Doctor rushed out to make production for the day and in passing he ha said drink more water and lose weight which have been doing. I has surgery on right kidney 48 years ago to correct congenital defect blocked urethral pelvic junction. Had stone in that kidney 25 years and passed naturally.
Expectations for future stones there
Thank you so much for any advice you can provide
Philip Brandt
Hi Philip, Because of the past surgery the right kidney may be specially vulnerable and I presume the new one was in that kidney. The composition of the stone is important because in older people uric acid is common and it is a totally preventable kind of stone. A full evaluation is needed if the stone is simply calcium based, and treatment proceeds from what is found. Without attention, another stone may form, but prediction of isolated events in one person is naturally fraught. Regards, Fred Coe
Hi Dr Cole,
Thank you for the very informative information. I’d been having some vague symptoms for the last year or two (mainly tiredness and lack of energy), Eventually this came to a head on Aug 15 this year when Some Calcium stones partially blocked one of my kidneys, it turns out that I had one 8mm, one 6mm and one 1mm Calcium stones in my left kidney and one 1mm stone in my right kidney. I had the two large stones in my left kidney broken up by laser in August, and had a stent in place for a few weeks. Apart from the discomfort of the stent, everything went well, and I was no longer tired and had loads of energy.
However, since early October, I’ve been aware of my left kidney feeling “not right”. My first x-ray after having the stents removed, showed a “vague mass” in my left kidney, which was thought to be some “sand” left by the procedure. A subsequent ultrasound showed a huge, (literally a boulder) in my left kidney, which was dismissed as the ultrasound technician “getting it wrong”.
However this last weekend, I showed symptoms of having another, temporary blockage of my kidney, with the same symptoms as I had last time, including dry heaving etc.
My questions is, how likely/common is it for stones to come back so soon after initial treatment? I had them originally removed just over 3 months ago? Is this normal? Or could there be another reason for their quick regeneration?
Thank you!
Hi Stuart, Stones can re-grow rapidly, in some instances. If the ultrasound is in doubt an ultra low dose CT will settle things. It is highly accurate and delivers very little radiation. Your physician might want to consider one. An alternative is that sand was indeed left behind and is passing with symptoms. That is not re-growth and should gradually abate. Regards, Fred Coe
Dear Dr. Coe,
Thank you for making this information available online. I am always curious about how the human body works (and fails to work), and have never been satisfied just getting a diagnosis. As I am currently in the throes of passing my first kidney stone, that curiosity is hyper-focused on what really goes on in the kidney, how things can go wrong, and how to avoid those problems. What a delight to happen upon your work here! It is always encouraging to see medical professionals truly unlocking all the secrets and nuances of the human body’s systems, moreover to find them making that knowledge plainly and publicly available; in an age where most of our contact with the profession involves little more than having our symptoms matched to pharmaceuticals. Thank you and may your efforts return to you many times over.
Cheers,
Mary
Hi Mary, I am glad the site has helped you. Be sure and read Chapter One, which is about prevention. Very important. Regards, Fred Coe
Thx for your information on kidney stones I drink mostly petite water which contains 6 percent 700 mg of calcium per bottle several per day This is the second time I will be needing surgery Do you think Drinkig this water would contribute to kidney stones ? Thank you for your input Paul
Hi Paul, Are you saying the water contains 700 mg calcium /bottle and you use several bottles a day = 1400 mg calcium? That is a lot and also possibly being taken away from meals. Of course your stones may be from many causes, and you need proper testing, but the water is a bit suspicious.
Hi,
I wondered why some stones pass when they are small – for example I have passed numerous 5mm stones, but others continue to grow? I have had surgeries for a 12mm stone a few years ago. This year, I had an 18mm stone per a CT scan in March and by my surgery date in August it had grown to 30mm. Why does the body push some out before they reach that size yet others linger and grow? My stones are carbonate apatite if that matters. Thank you!
Hi Lucy, That stones are carbonate apatite is crucial. These form only in alkaline urine and point to where you may have abnormalities causing stones. Drugs can do this, often diet, or inheritance. Urine calcium is often high. Treatment depends on your testing, so be certain you have been evaluated well – Rapid growth points to important abnormalities not being treated. Regards, Fred Coe
Good Morning Dr. Coe,
This was a very interesting read.. thank you. I currently have a 6 mm kidney stone and two smaller ones that are being blocked from passing. I am awaiting surgery to “blast them” and currently have a stent in for 3 weeks which has been very painful. I have so many reasons that could be why I have the stones, (dehydration, 2 years of on again off again antibiotics due to infections from breast cancer surgery, overweight, keto diet, major tea drinker) but I keep looking for how long it can take to create the kidney stones in your body. I have looked on the internet to no avail. Just trying to figure that part out and it will help me to potentially understand why I created this monster.
Thank you for any insight on this and for all you are doing to help us understand this extremely painful medical problem.
Traci
Can you please share how long it takes to create…months.. years…
Thank you!!
Hi Traci, It depends on calcium oxalate vs calcium phosphate. The latter grow faster – months to years, the former many months to years. But in a general sense, it is possible you produced the nuclei for stones during dehydration and diet changes which grew into the stones. Whatever the timing, I would advise you get fully evaluated now, and treat whatever is wrong. Regards, Fred Coe
Dr. Coe,
Thank you for your response to my question! At this time I don’t know what type of Kidney stones I have, other than the size.
Is it possible to grow a 6mm kidney stone in a couple of months? The reason I ask, I had deep flap surgery in November and was very dehydrated due to the difficulty in moving around. (not wanting to drink and have to use the restroom because it was too painful) Not only did I have the surgery, but developed a very large wound requiring a wound vac most likely till April.
The kidney stone episode happened two months after November surgery, with significant pain, hospital stay, stent surgery and awaiting the shockwave lithotripsy surgery later this week. I will have the stent for a total of 2 1/2 months due to Dr scheduling.
If a 6mm kidney stone can develop that quickly, (2 months) then I will make some assumptions about why this happened to me. Thank you again for your kindness in answering my question.
Hi Traci, Given the history I would be concerned the stones are calcium phosphate or uric acid, which grow very rapidly. Analysis of removed stone material is crucial for treatment, as is 24 hour urine testing. Regards, Fred Coe
Hi Dr. Coe,
I’m trying to determine if my first kidney stone is a result of genetics (my father has kidney stones) or a side effect of a medication (topiramate). The kidney pain began and above 19 days after I began the medication. Is it possible for a stone to form that quickly? My doctor doesn’t think so but didn’t sound confident and I wanted a second opinion.
Hi Abigail, Topiramate is really a stone forming drug – calcium phosphate stones. 19 days is short, but the drug is a suspect. Stop it, of course, and get another more safe agent from your personal physicians, one that does not inhibit carbonic anhydrase. Then, get a proper evaluation for causes of stones – here is a good introduction to how. Regards, Fred Coe
My stone is 8mm and the first urologist had nicked the ureter trying to get to it. I was told it is embedded in the kidney wall. A new surgeon with a better skill set is now set to attempt to take it out. Mind you I’ve never had symptoms, and they only found it from a contrast because of another unrelated hospital stay. My question is does it need to come out? What implications are there if it stays? They make it sound as if they are only going to get they can and then monitor me every 6 months. By the way the first surgeon did leave the stent in , so I’m wondering if I can just have that taken out? Thanks in advance
Hi Jim, Complicated. If the stone is embedded in the kidney tissue, most folks would leave it alone unless it causes obstruction, infection, pain, or serious bleeding. As for the ureter, the stent is probably left in place to foster healing. As for a second attempt to remove it, perhaps there are surgical indications I do not know about, perhaps the ureter needs to heal up or will need special care. If your new surgeon has the needed skills, it is up to him/her to use them and fix things. I guess I do not have enough facts to add anything more. Fred
Hello. I found this page, and was very interested. Perhaps it can explain my problem. I am 64 years old and had my first stone when I was 23. i have had one stone analyzed, it was a calcium stone. In my left kidney I have a stone 9 mm, it has been there for ages and caused my no trouble. But for the last two weeks I have had a lot of stone attacs. They Are very painfull, some of them so painfull that I wommit. But they last for some hours, and then it is over. I can follow the pain from my back down the urinetrackt. But I cannot see any stones, only tiny small grains. Tiny tyny. But it wont stop, they just come and come. Have had at least Ten now I think. What can this be? Will this go on snd on? What can I do to stop it? Can it come from the big stone indide the kidney. i have been to hospital to remove stones, but I do not get any good follow up. i am really quit desperate now…I take calcium Chanel blockers for my blood pressure. Can they cause it? I eat a low sodium and low oxalate diet. Best whishes from Rikke in Norway
Hi Rikke, You are describing passage of crystals – too small to be stones, easily caught on filter paper – like a coffee filter – and analyzed by infrared spectrum. If I had to guess at are 64 it would be uric acid – sometimes visible as orange or red particles. Your physician can look at the urine under a microscope for crystals – they can be told apart from their morphology if you cannot collect some on a filter. So the first issue is to know what is passing, then prevention is rather simple. Regards from Fred in Lakeside Michigan.
Thank you for this article. I am a 50 yr old female who was diagnosed with MSK after several years of reoccurring calcium stones varying from 2.5 mm to 8 mm. I am currently suffering from an 8 mm stone identified by the ER via CT and will be following up with my urologist shortly. I have been told MSK is a congenital disease and recently was advised to consult an ENT specialist for a possible parathyroid tumor that may be contributing to increased blood calcium levels. Visual inspection of the in office sonogram by the ENT points to a likely benign tumor but I will undergo additional testing to confirm. I’m interested in your thoughts about the connection between the parathyroid and calcium stone formation and whether addressing the tumor (if there is indeed one) will “cure” my frequent calcium stones. I am extremely healthy otherwise. I don’t smoke, don’t consume alcohol, am allergic to sugar cane and corn and any derivatives so my diet is better than most, and I am of a healthy weight. I appreciate your time and eagerly await your response.
Hi Tamy, primary hyperparathyroidism is a prime cause of stones and almost invariably curable by surgical removal of the offending gland(s). The surgery is very specialized and should be performed by an endocrine surgeon. Almost always the disease is benign and curable. The article details diagnosis and possible pitfalls. Regards, Fred Coe
Hi, Fred, Thanks so much for all the information on kidney stones! I’ve had two surgeries to remove calcium oxalate stones larger than a centimeter and just learned I have another 5 mm one. My doctor said this is passable but that it’s likely still stuck to the wall of the kidney. Is there a chance it can continue to grow to a size that is not passable if left long enough? And is there anything I can do other than drink water to help dissolve/dislodge and pass it? After the first surgery, I significantly increased fluid intake and worked with a dietician so don’t eat a diet conducive to forming the kinds of stones I get. The thought of having to endure another surgery and stents makes me desperate to try anything. Thank you!
Hi Bob, Prevention depends on the cause of the stones. Here is a good plan for a full evaluation. Be sure you have had what it takes and look at the results with your physician to see what is abnormal and causing stones. Treatment with water and diet sans knowledge of the real cause is not likely to work well. Regards, Fred Coe